Update on the spondyloarthropathies
The spondyloarthropathies (SpA) are strongly associated with the HLA-B27 gene. The diagnosis is based primarily on clinical findings. Ankylosing spondylitis (AS) often involves the sacroiliac joints and spine. Psoriatic arthritis (PsA) occurs in up to about one third of patients with psoriasis. Reactive arthritis must be distinguished from other arthropathies. Arthritis occurs in about 30% of patients with inflammatory bowel disease. Undifferentiated SpA include sseveral related disorders. Radiographic evidence of sacroiliitisis a characteristic feature of AS. SpA management should include patient education and regular exercise. NSAIDs are the first line of treatment.The tumor necrosis factor α inhibitors are highly effective inpatients with active AS and in those with PsA that is unresponsive to conventional therapy. (J Musculoskel Med. 2008;25:31-40)
ABSTRACT: The spondyloarthropathies (SpA) are strongly associated with the HLA-B27 gene. The diagnosis is based primarily on clinical findings. Ankylosing spondylitis (AS) often involves the sacroiliac joints and spine. Psoriatic arthritis (PsA) occurs in up to about one third of patients with psoriasis. Reactive arthritis must be distinguished from other arthropathies. Arthritis occurs in about 30% of patients with inflammatory bowel disease. Undifferentiated SpA includes several related disorders. Radiographic evidence of sacroiliitis is a characteristic feature of AS. SpA management should include patient education and regular exercise. NSAIDs are the first line of treatment. The tumor necrosis factor α inhibitors are highly effective in patients with active AS and in those with PsA that is unresponsive to conventional therapy. (J Musculoskel Med. 2008;25:31-40)
Together the spondyloarthropathies (SpA) form a group of overlapping chronic inflammatory rheumatologic diseases that show a predilection for involvement of the axial skeleton, entheses (bony insertions ofligaments and tendons), and peripheral joints. They also may involve extra skeletal structures, especially the eyes, lungs, skin, and GI tract. These diseases are strongly associated with the HLA-B27 gene but lack association with rheumatoid factor (RF) and antinuclear antibodies.1
The SpA include ankylosingspondylitis (AS), psoriatic arthritis (PsA), reactive arthritis (ReA), arthritis of inflammatory bowel disease (IBD), and undifferentiated SpA. They are more common than previously recognized. Recent data from Europe and Asia suggest that as a group, the SpA might be as common as rheumatoid arthritis (RA); in Europe, the prevalence is 0.5% to 1%.2-4
Because there are no diagnostic criteria for the wider spectrum of the SpA, the diagnosis is based primarily on clinical findings.1,2,4-8 European Spondylarthropathy Study Group (ESSG) classification criteria are used frequently to help the clinical diagnosis (Table 1).9 Early diagnosis has become much more important in recent years as more effective therapeutic options have become available.

In this article, we describe the specific clinical entities in the SpA and their common laboratory and radiological features. Then we outline a variety of management strategies, including nonpharmacological modalities, pharmacological therapy, and ophthalmological or surgical referral.
DIAGNOSIS
Clinical manifestations
AS. The prototype of the SpA, AS primarily involves the sacroiliac joints and spine (Figure) and, often, the hip and shoulder joints; patients typically present with chronic inflammatory back pain.2,8 Symptoms usually start insidiously when patients are in their late teens or early 20s; men are affected roughly twice as frequently as women.
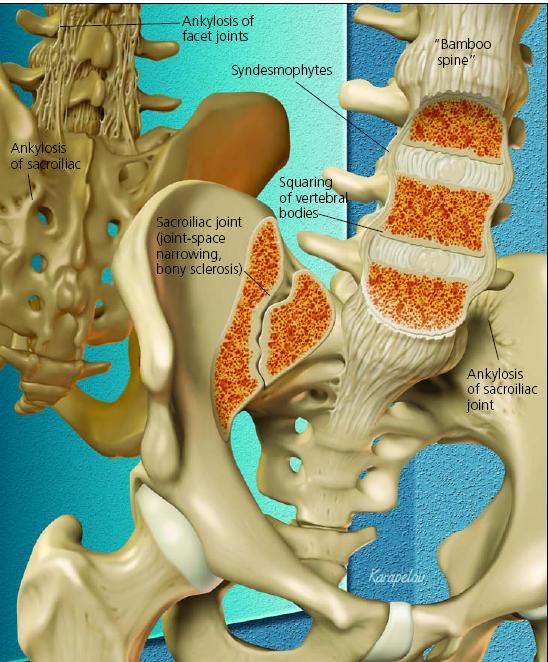
Figure – Ankylosing spondylitis, the prototype of the spondyloarthropathies, involves the sacroiliac joints and spine. Radiographic evidence of sacroiliitis, usually bilateral and symmetrical, is a characteristic feature. Spinal involvement may lead to squaring of vertebral bodies; formation of syndesmophytes, erosions and, later, ankylosis of facet joints; ligament ossification; and ultimately, “bamboo spine.”
Patients with AS may awaken late at night or very early in the morning because of back pain and stiffness, which is eased with physical exercise or a hot shower. Enthesitis may cause pain and tenderness over the anterior chest wall, spinal processes, iliac crests, and sites of bony insertions of the Achilles and patellar tendons and plantar fascia. Peripheral arthritis, usually monoarticular or oligoarticular, is less common in primary AS than in “secondary” AS (in the context of PsA, ReA, or IBD).
Tenderness may be noted over the sacroiliac joints, spinal processes, and other bony prominences. In some patients, pain in the sacroiliac joint area may be elicited with sacroiliac stress testing by using maneuvers such as the FABERE test (hip Flexion, Abduction, External Rotation, and Extension).8 Costovertebral and costotransverse joint involvement may result in diminished chest expansion. Gradual impairment of spinal mobility maybe noted on lateral and forward flexion, hyperextension, and axial rotation of the lumbar and cervical spine. Limitation of forward flexion of the lumbar spine may be measured with the modified Schober test; occiput-to-wall distance measures the forward stooping deformity of the neck.8
Acute anterior uveitis, the most common extra skeletal manifestation of AS, occurs in up to 40% of patients with AS, especially those who possess the HLA-B27 gene.1,2,8 If not recognized and managed early, uveitis may lead to visual impairment. Subclinical lung abnormalities are somewhat common inpatients with AS10; however, clinical pulmonary manifestations are uncommon. Other, less common extra skeletal manifestations of AS may involve the gut, aorta, or heart. Cauda equina syndrome is a rare neurological complication of AS.1,2,8
In the absence of diagnostic criteria for AS, the modified New York criteria are the most commonly used classification criteria (Table 2).11 These criteria are highly specific rather than sensitive and are used mainly for including patients in clinical studies.

PsA. This inflammatory arthritis occurs in 10% to 30% of patients with psoriasis, which may not be readily apparent because psoriasis lesions may be limited to the scalp, ears, umbilicus, perineum, and perianal area. Therefore, a thorough skin examination should be performed in every patient with inflammatory arthritis.1
The onset of arthritis usually follows or coincides with the onset of psoriasis, although it may antedate psoriasis in up to 15% of patients. Arthritis may present in various overlapping forms, including polyarthritis, asymmetrical oligoarthritis (involving fewer that 5 joints), arthritis primarily limited to the distal interphalangeal (DIP) joints, monarthritis, arthritis mutilans, sacroiliitis, and spondylitis. The polyarthritis form resembles RA but has several features quite characteristic of PsA, including the involvement of the DIP joints; the presence of dactylitis, or “sausage digits,” and enthesitis; and nail involvement (discoloration, onycholysis, ridging, and especially, pitting). Psoriatic spondylitis is clinically similar to AS but more often associated with peripheral arthritis and less often with uveitis.
ReA. This condition classically occurs within 1 to 4 weeks after a triggering infection of the gut or the genitourinary tract, although many patients may not recall such a history.7 Patients with ReA usually present with acute, asymmetrical oligoarthritis of the lower extremities. They also may have constitutional symptoms, urethritis, cervicitis, conjunctivitis, uveitis, genital lesions (circinate balanitis or vulvitis), keratoderma blennorrhagica, dactylitis, enthesitis, sacroiliitis/spondylitis, nail discoloration and onycholysis without nail pitting, or sacroiliitis/spondylitis. The uncommon classic triad of arthritis, conjunctivitis, and urethritis maybe present in a subset of patients with ReA.
ReA must be distinguished from other arthropathies that present in a similar fashion, especially PsA, septic arthritis, gout, pseudogout, juvenile arthritis, and sarcoid arthropathy. Poststreptococcal ReA has been described in patients who do not meet the classification criteria for rheumatic fever. HIV infection has been strongly associated with ReA, PsA, and undifferentiated SpA in sub-Saharan populations in Africa.12
Arthritis of IBD. Arthritis occurs in about 30% of patients who have IBD; arthritis of IBD manifests with inflammatory back pain, enthesitis, or peripheral arthritis, fulfilling the ESSG criteria for SpA; 10% of patients fulfill the criteria for AS.13 In addition, some patients have asymptomatic sacroiliitis. Peripheral arthritis usually is nonerosive and, unlike axial disease, correlates with the IBD activity.
Undifferentiated SpA. This encompasses related disorders that include such clinical entities as isolated enthesitis or dactylitis and RF-negative oligoarthritis or polyarthritis; it usually involves the lower extremities and often is HLAB27-associated.1 Patients who have undifferentiated SpA may have episodes of acute anterior uveitis with 1 of the above-mentioned features but not psoriasis or GI or genitourinary tract involvement.
Laboratory features
The C-reactive protein (CRP) level and erythrocyte sedimentation rate (ESR) often are elevated with peripheral joint involvement in patients with SpA, but they may be normal in many patients, especially those who have isolated axial disease. Anemia of chronic disease might occur. Synovial fluid analysis shows inflammatory changes that are nonspecific; the results of bacterial studies typically are negative, although the studies should be performed to rule out septic arthritis. The results of a fecal occult blood test may be abnormal in patients with IBD. When ReA is suspected on clinical grounds, bacterial studies (eg, throat cultures and tests for urogenital and enteric infection) might provide helpful information, although they may not be necessary for making the diagnosis.7 HIV testing should be considered in high-risk patients.
Testing for the HLA-B27 gene may help in some clinical situations (eg, when the clinician estimates that the pretest disease probability is about 50% and the radiographic evidence for sacroiliitis is equivocal or absent).1,8HLA-B27 testing cannot be used for screening or as a routine test for making a diagnosis of AS and related SpA because these diseases may occur in the absence of this gene and it may be present in a sizable percentage of healthy persons.1,8
Radiological features
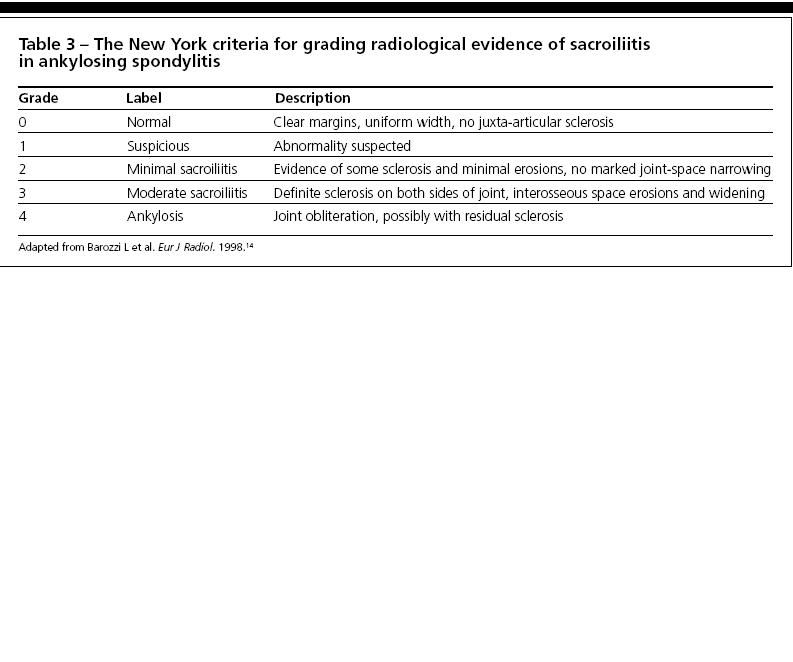
Radiographic evidence of sacroiliitis is a characteristic feature of AS; it usually is bilateral and symmetrical in primary AS and typically first involves the lower synovial part of the joint (Table 3).14 However, sacroiliitis often is unilateral or asymmetrical in ReA and PsA.
Obtaining an anteroposterior x-ray film of the pelvis may be sufficient for detecting sacroiliitis, but the results may be normal or equivocal early in the disease course. MRI with short tau inversion recovery technique, without the need for gadolinium enhancement, may detect sacroiliitis or spondylitis long before these abnormalities become evident on plain x-ray films.15 This offers the clinician an opportunity for early diagnosis, especially with the availability of more effective treatment in recent years. If MRI is not available or cannot be used, CT scanning may be helpful.14
The recently described total body MRI scan may very nicely identify the characteristic bone edema that results from osteitis/enthesitis at axial as well as peripheral sites. It takes only 30 minutes to perform.15 Technetium bone scanning is not very helpful for early detection of sacroiliitis, but it may be used to detect pseudoarthrosis or fracture.16
Spinal involvement may gradually lead to the squaring of vertebral bodies, formation of syndesmophytes, erosions, and later, ankylosis of facet joints and ligament ossification that are characteristic of AS. These changes ultimately may result in a completely fused spine that resembles a bamboo stick, hence the name “bamboo spine” (see Figure). Atlantoaxial fusion or subluxation also may occur.
Spinal osteopenia, which is common, correlates with disease severity and duration. Measurements of the spine by dual-energy x-ray absorptiometry (DEXA) scan to detect osteoporosis may be less reliable than measurements at the femoral neck because of the presence of spinal syndesmophytes and ligamentous ossification. Peripheral DEXA scanning may be used in patients who have had bilateral hip arthroplasty.
Radiographic findings in patients with AS secondary to PsA or ReA may be asymmetrical, with skip lesions or nonmarginal, large, asymmetrical syndesmophytes or both. Radiographic features of PsA include erosions, periostitis, bony ankylosis, and destructive “pencil-in-cup” deformities (a bone’s distal head becomes pointed and the adjacent joint surface becomes cup-like because of erosions in the hands and feet). Periarticular osteopenia, often seen in RA, is a not a typical feature in the SpA.
Radiographic evidence of sacroiliitis, especially unilateral or asymmetrical, may result from other causes-including infections (eg, brucellosis and tuberculosis)-but the clinical presentation usually is sufficiently discernible from that of AS and related SpA. Osteitis condensans ilii (sclerosis of the iliac side of the sacroiliac joints without cortical erosions or joint-space changes) also must be distinguished from sacroiliitis in women, especially those with a history of multiple pregnancies.
Diffuse idiopathic skeletal hyperostosis (DISH), also known as ankylosing hyperostosis or Forestier disease, may be confused with AS but usually occurs in older patients. DISH is characterized by flowing ligamentous ossification, especially of the anterior longitudinal ligament, and the absence of typical sacroiliitis, although capsular ossification may diminish the clarity of the sacroiliac joints on roentgenographic images.17
MANAGEMENT
SpA management should be individualized according to the patient’s clinical presentation, comorbidities, and wishes. Guidelines have been published for AS management.18 The best strategy for AS incorporates patient and family education; lifestyle and workplace modifications; physical therapy; a lifelong program of regular physical exercise; use of medications; referral to appropriate specialists, when needed; and monitoring of disease activity and adverse effects of drug therapies.
Patient education
Patients should be provided with disease-specific written instructions and illustrations, handouts, books, pamphlets, videos, audiotapes, and information about useful Web sites. Self-help programs and patient education and counseling improve patients’ adherence with therapy regimens and benefit their general health and functional status.18,19 Cessation of cigarette smoking should be strongly encouraged because smoking is associated with worse outcomes; patients also should receive pneumococcal and yearly influenza vaccinations, especially when treatment with tumor necrosis factor α (TNF-α) inhibitors is being considered.
Patients’ posture problems and difficulties in performance of activities of daily living should be identified, and workplace modifications may be needed. Frequently changing position when sitting at a desk and taking breaks for body stretching are helpful. Activities that cause back muscle strain should be avoided (eg, prolonged stooping or bending and assuming positions that may cause a stooped posture, such as prolonged slouching in chairs or leaning over a desk).
Helpful assistive devices include long-handled devices for dressing and for reaching or picking up objects, adjustable swivel chairs that provide lumbar support, and elevated and inclined writing surfaces. Use of wide-view mirrors helps driving for patients who have limited neck mobility. Use of back splints, braces, and corsets is not helpful and should be avoided.
Exercise and physical therapy
A lifelong program of regular exercise should be encouraged; such a program should include spinal extension exercises; deep breathing; and range of motion exercises of the back, neck, shoulders, hips, and other joints.19 Swimming and aquatic exercises and other appropriate recreational exercises are especially useful. High-impact and contact sports activities and those that involve abrupt movement of the spine should be avoided, especially by patients who have axial disease with limited spinal mobility.
Short-term intensive physical therapy followed by lifelong home exercises might be helpful for patients with SpA, including core strengthening of trunk muscles. However, the role of physical therapy has not been assessed since TNF-α inhibitors became available.
Pharmacological treatment
NSAIDs given at the full anti-inflammatory dose are the first line of treatment for patients with the SpA1,2,20; it is better to take them regularly.21 Because of variation of response, some patients may need to be switched to other NSAIDs to find the most effective one before giving up on NSAIDs for disease management.
The traditional disease-modifying antirheumatic drugs-eg, methotrexate (MTX), leflunomide, and sulfasalazine-are not recommended for managing axial disease because they lack efficacy. However, they may be considered in patients with peripheral arthritis. Oral corticosteroids should be avoided, but intra-articular or local corticosteroid injection may provide rapid relief in monarticular or oligoarticular peripheral arthritis or enthesitis.
Osteoporosis is quite common in patients with SpA and should be recognized and managed early; adequate intake of calcium and vitamin D should be ensured. Patients with Chlamydia-induced ReA and sexual partners may need to be treated with appropriate antibiotics simultaneously to more effectively eradicate the infection, although such treatment may not alter the natural disease course.1,7
All 3 TNF-α inhibitors-infliximab, etanercept, and adalimumab-are highly and equally effective in patients with active AS and in those with PsA and enteropathic arthritis that is unresponsive to conventional therapy.22-27 These biologic agents also are very effective in managing the cutaneous and nail lesions of psoriasis.2,25-27 Infliximab and adalimumab (monoclonal antibodies) are effective in managing IBD, but etanercept (receptor-fusion protein) lacks such an effect. The monoclonals also are somewhat more effective in preventing recurrences of acute anterior uveitis.
The TNF-α inhibitors are effective as monotherapy without concomitant MTX, and they have maintained long-term effectiveness.2,18,28 Clinical improvement is accompanied by a significant decrease in inflammation, as evidenced by a dramatic reduction of the CRP level and ESR; the improvement also may be demonstrated on MRI, but it is too early to say that these agents will slow down or prevent progressive bony ankylosis.2,28 A few patients with ReA and with undifferentiated SpA that is refractory to traditional therapies who have been treated with TNF-α inhibitors also have shown good response.29
Guidelines for the use of TNF-α inhibitors for the SpA have been developed (Table 4).18,30 Treatment must be continued on a long-term basis to maintain disease control. When one TNF-α inhibitor has not succeeded or adverse effects develop that are not related to the TNF-α inhibitors as a class, switching to another agent may be indicated. The TNF-α inhibitors are expensive and may be associated with potentially serious adverse effects, including allergic and injection-site reactions, and an increased risk of infections, including reactivation of tuberculosis.
REFERRALS
In cases of acute anterior uveitis, ophthalmological evaluation is urgently needed. In cases of other extraskeletal complications, consider referral to a cardiologist, pulmonologist, or other specialist.
In patients of all ages who have advanced hip or knee joint damage with intractable pain or disability or both, surgical referral should be considered for total joint replacement. The risk of heterotopic bone formation around the prosthesis may be reduced by use of NSAIDs for 7 to 10 days from the day of surgery.31 Any new-onset neck or back pain in a patient with AS should be evaluated carefully for a spinal fracture or instability, even in the absence of trauma or after a seemingly trivial injury, because paraplegia or quadriplegia may result. If fracture is suspected, the neck should be immobilized as a precaution pending musculoskeletal imaging. The x-ray findings may be normal, but MRI, CT, or bone scan results may be more helpful in confirming or excluding spinal fracture.
Elective spinal surgeries that might be indicated include osteotomy to correct severe kyphosis and uncompensated loss of horizontal vision and fusion procedures for instability, including atlantoaxial subluxation, pseudoarthrosis, and fracture. General anesthesia is associated with intubation difficulties in cases of cervical spine ankylosis and deformity and involvement of the temporomandibular joint. The risk of pulmonary complications increases during the postoperative period, and special care is required.
References:
References
- 1. Khan MA. Update on spondyloarthropathies. Ann Intern Med. 2002;136:896-907.
- 2. Braun J, Sieper J. Ankylosing spondylitis. Lancet. 2007;369:1379-1390.
- 3. Akkoc N, Khan MA. Epidemiology of ankylosing spondylitis and related spondyloarthropathies. In: Weisman MH, Reveille JD, van der Heijde D, eds. Ankylosing Spondylitis and the Spondyloarthropathies: A Companion to Rheumatology. London: Mosby-Elsevier; 2006:117-131.
- 4. Khan MA, Clegg DO, Deodhar AA, et al. 2006 Annual research and education meeting of the Spondyloarthritis Research and Therapy Network (SPARTAN). J Rheumatol. 2007;34:1118-1124.
- 5. Rudwaleit M, Khan MA, Sieper J. The challenge of diagnosis and classification in early ankylosing spondylitis: do we need new criteria? Arthritis Rheum. 2005;52:1000-1008.
- 6. Elyan M, Khan MA. Early diagnosis of ankylosing spondylitis. J Rheumatol Suppl. 2006;78:12-23.
- 7. Khan MA, Sieper J. Reactive arthritis. In: Koopman WJ, Moreland LW, eds. Arthritis and Allied Conditions. 15th ed. Philadelphia: Lippincott Williams and Wilkins; 2004:1335-1355.
- 8. Khan MA. Ankylosing spondylitis: clinical features. In: Hochberg M, Silman A, Smolen J, et al, eds. Rheumatology. 3rd ed. London: Mosby; 2003:1161-1181.
- 9. Dougados M, van der Linden S, Juhlin R, et al. The European Spondyloarthropathy Study Group preliminary criteria for the classification of spondyloarthropathy. Arthritis Rheum. 1991;34:1218-1227.
- 10. Sampaio-Barros PD, Cerqueira EM, Rezende SM, et al. Pulmonary involvement in ankylosing spondylitis. Clin Rheumatol. 2007;26:225-230.
- 11. van der Linden S, Valkenburg HA, Cats A. Evaluation of diagnostic criteria for ankylosing spondylitis: a proposal for modification of the New York criteria. Arthritis Rheum. 1984;27:361-368.
- 12. Mijiyawa M, Oniankitan O, Khan MA. Spondyloarthropathies in sub-Saharan Africa. Curr Opin Rheumatol. 2000;12:281-286.
- 13. de Vlam K, Mielants H, Cuvelier C, et al. Spondyloarthropathy is underestimated in inflammatory bowel disease: prevalence and HLA association. J Rheumatol. 2000;27:2860-2865.
- 14. Barozzi L, Olivieri I, De Matteis M, et al. Seronegative spondylarthropathies: imaging of spondylitis, enthesitis and dactylitis. Eur J Radiol. 1998;27(suppl 1):S12-S17.
- 15. Weber U, Pfirrmann CW, Khan MA. Ankylosing spondylitis: update on imaging and therapy. Int J Adv Rheumatol. 2007;5:2.
- 16. Miron SD, Khan MA, Wiesen EJ, et al. The value of quantitative sacroiliac scintigraphy in detection of sacroiliitis. Clin Rheumatol. 1983;2:407-414.
- 17. Yagan R, Khan MA. Confusion of roentgenographic differential diagnosis between ankylosing hyperostosis (Forestier’s disease) and ankylosing spondylitis. Clin Rheumatol. 1983;2:285-292.
- 18. Zochling J, van der Heijde D, Burgos-Vargas R, et al. ASAS/EULAR recommendations for the management of ankylosing spondylitis. Ann Rheum Dis. 2006;65:442-452.
- 19. Elyan M, Khan MA. Spondyloarthropathies. In: Bartlett SJ, Bingham CO, Maricic MJ, et al, eds. Clinical Care in the Rheumatic Diseases. 3rd ed. Atlanta: Association of Rheumatology Health Professionals; 2006:177-185.
- 20. Elyan M, Khan MA. The role of nonsteroidal anti-inflammatory medications and exercise in the treatment of ankylosing spondylitis. Curr Rheumatol Rep. 2006;8:255-259.
- 21. Wanders A, van der Heijde D, Landewe R, et al. Inhibition of radiographic progression in ankylosing spondylitis (AS) by continuous use of NSAIDs. Arthritis Rheum. 2005;52:1756-1765.
- 22. van der Heijde D, Dijkmans B, Geusens P, et al. Efficacy and safety of infliximab in patients with ankylosing spondylitis: results of a randomized, placebo-controlled trial (ASSERT). Arthritis Rheum. 2005;52:582-591.
- 23. Davis JC Jr, Van Der Heijde D, Braun J, et al; Enbrel Ankylosing Spondylitis Study Group. Recombinant human tumor necrosis factor receptor (etanercept) for treating ankylosing spondylitis: a randomized, controlled trial. Arthritis Rheum. 2003;48:3230-3236.
- 24. van der Heijde D, Kivitz A, Schiff MH, et al. Efficacy and safety of adalimumab in patients with ankylosing spondylitis: results of a multicenter, randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 2006;54:2136-2146.
- 25. Antoni C, Krueger GG, de Vlam K, et al. Infliximab improves signs and symptoms of psoriatic arthritis: results of the IMPACT 2 trial. Ann Rheum Dis. 2005;64:1150-1157.
- 26. Ory P, Sharp JT, Salonen D, et al. Etanercept (ENBREL) inhibits radiographic progression in patients with psoriatic arthritis. Arthritis Rheum. 2002;46(suppl 9):S196.
- 27. Mease PJ, Gladman DD, Ritchlin CT, et al. Adalimumab for the treatment of patients with moderately to severely active psoriatic arthritis: results of a double-blind, randomized, placebocontrolled trial. Arthritis Rheum. 2005;52:3279-3289.
- 28. Baraliakos X, Listing J, Rudwaleit M, et al. Radiographic progression in patients with ankylosing spondylitis after 2 years of treatment with the tumour necrosis factor alpha antibody infliximab. Ann Rheum Dis. 2005;64:1462-1466.
- 29. Meador R, Hsia E, Kitumnuaypong T, Schumacher HR. TNF involvement and anti-TNF therapy of reactive and unclassified arthritis. Clin Exp Rheumatol. 2002;20(6 suppl 28):S130-S134.
- 30. Spondylitis Association of America. Guidelines for the use of anti-TNF therapy in patients with ankylosing spondylitis. 2007. Available at: http://www.spondylitis.org/physician_ resources/guidelines.aspx. Accessed December 4, 2007.
- 31. Nilsson OS, Persson PE. Heterotopic bone formation after joint replacement. Curr Opin Rheumatol. 1999;11:127-131. Â