Eradication of Hepatitis C: High Hopes, But Challenges Remain
Hepatitis C virus (HCV) has been referred to as a "stealth virus" because most people who develop chronic hepatitis C don't know they are infected.

Hepatitis C virus (HCV) has been referred to as a “stealth virus” because most people who develop chronic hepatitis C don’t know they are infected. Infected individuals can remain asymptomatic for decades until HCV’s typically slow progression causes destructive complications—cirrhosis, liver failure, or hepatocellular carcinoma (HCC).1 The spread of HCV has been termed a “silent epidemic,” and the World Health Organization describes the growing prevalence of this common, blood-borne infection as a “viral time bomb.” Silent no more, HCV now has the spotlight, for both positive and negative reasons.
On the negative side, the “viral time bomb” is expected to explode soon, very soon. Of the almost 4 million Americans infected with HCV, 50% to 75% are chronically infected but still unaware of their condition; they are just beginning to experience the most serious effects of the virus.1-4 In the United States, liver cancer has most often affected older adults (mean age 65), but that is changing. Currently, incidence is shifting to younger people, primarily affecting men aged 45 to 65.2 Prior to 1990, many were infected before blood banks knew how to screen for the virus, and others were infected during the 1960’s, 70’s, and 80’s as HCV infection paralleled a rise in illicit drug use and shared needles.

A study released in February 2012 reported that in 2007, for the first time, HCV-related deaths in the United States surpassed those caused by HIV.4 Another study showed that liver cancer has become the fastest growing cause of cancer-related death in the United States.5 Worldwide, chronic infection with hepatitis is the cause of up to 80% of liver cancers.4 In 2010, the American Cancer Society reported 24,120 cases of liver cancer, and in the same year, liver cancer caused 18,910 deaths.6
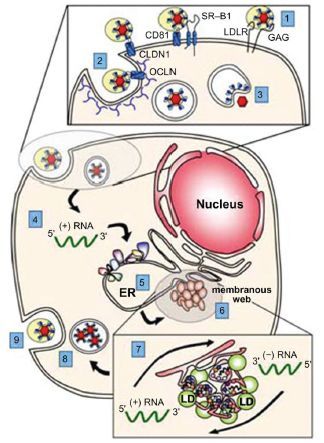
The positive news about HCV, however, is very good indeed. Dozens of new therapies for HCV are in the pipeline, and many are expected to come to market within the next two to five years. Better yet, the first two direct acting antivirals (DAAs) were approved in 2011. Boceprevir and telaprevir, the new agents, significantly increased the percentage of patients able to achieve a sustained viral response (SVR), which is the most reliable sign of a successful “cure.” The DAA agent was combined in a triple-therapy regimen with the existing standard of care (pegylated interferon and ribavirin). Thousands of HCV patients were treated in 2011 with one of the two new regimens, and outcomes continued to be positive.7
References
1. The “Secret” Epidemic: Disparities in Hepatitis C Incidence, Treatment and Outcomes. Joint Center for Political and Economic Studies. 2010.
2. Armstrong GL, et al. Ann Intern Med. 2006;144:705-714.
3.United Press International. Baby Boomers at Elevated Hepatitis C Risk. Available at: http://www.upi.com Health_News/2012/02/21/Baby-boomers-at-elevated-hepatitis-C-risk/UPI-45321329871972/.
4. Ly KN, et al. Ann Intern Med. 2012;156:271-278.
5. El-Serag, HB. N Engl J Med. 2011;365:1118-1127.
6. American Cancer Society. Cancer Facts & Figures. Available at: http://www.cancer.org/Research/CancerFactsFigures/index.
7. Alter HJ, Liang TJ. Ann Intern Med. 2012;156:317-318.
