FDA Approves AstraZeneca's Diabetes Injection Pen
A once-weekly self-injection pen has been approved by the FDA to supplement diet and exercise for type 2 diabetes patients.
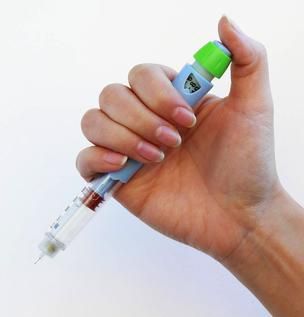
AstraZeneca announced its Bydureon Pen (exenatide extended-release for injectable suspension) 2 mg was approved by the US Food and Drug Administration (FDA). The pen is intended as a supplement to diet and exercise to improve glycemic control in adults with type 2 diabetes.
The once-weekly treatment is a pre-filled, single-use pen injector, which eliminates the need for patients to transfer the medication to a syringe during the self-injection process. Exenatide is delivered via microsphere technology that does not require titration, and can be administered any time of day, with or without meals.
“We’re pleased to receive approval for the Bydureon Pen, which can provide a powerful reduction in blood glucose levels along with the potential benefit of weight loss, through a once-weekly dose in a pre-filled device,” said Briggs Morrison, AstraZeneca Executive Vice President, Global Medicines Development and Chief Medical Officer. “We are committed to addressing the needs of adults with type 2 diabetes, including ongoing research to develop new treatments and methods of delivery.”
The pen contains the same formula and dose as the original Bydureon single-dose tray, which has been shown to provide blood glucose level reduction. The original medication was FDA-approved in 2012 and is currently on the market in 42 countries worldwide.
The new pen will come with the same warnings as the original formula, including risk of thyroid C-cell tumors, medullary thyroid cancer, potential increased risk of pancreatitis, fatal and nonfatal hemorrhagic, and necrotizing pancreatitis.
Bydureon is not a substitute for insulin or intended for patients with type 1 diabetes.