Retinopathy in Systemic Lupus Increases with Longer Hydroxychloroquine Use
Hydroxychloroquine retinopathy is more common than previously thought, shows a study published in Arthritis and Rheumatology. Higher dosages and long duration of use is associated with a higher risk of vision loss.
Diabetes Retinopathy (©AdobeStock_Koolsabuy)
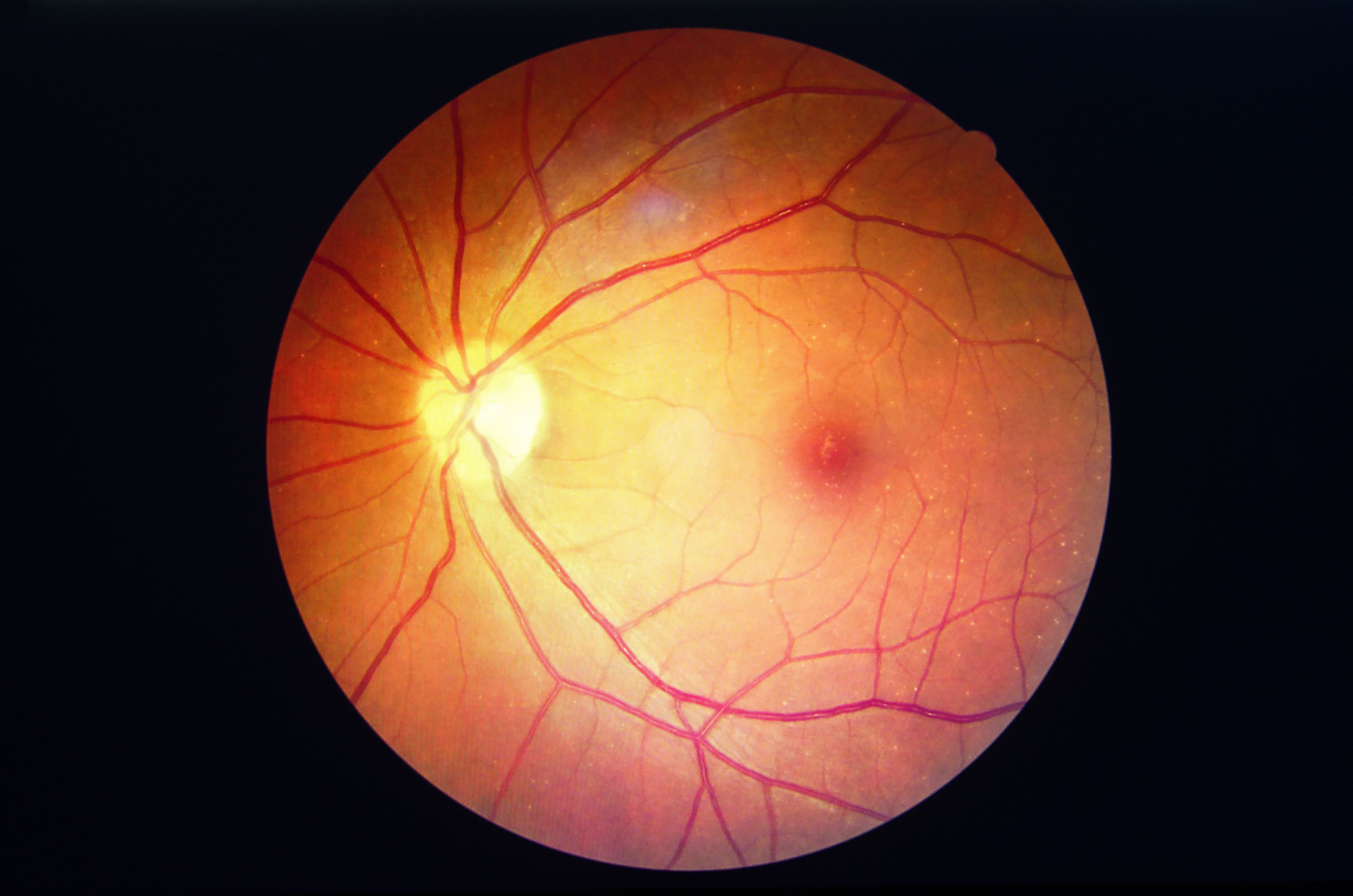
The incidence of retinopathy among systemic lupus erythematosus (SLE) patents treated with hydroxychloroquine increased with longer treatment duration, but could be predicted by monitoring blood levels of the drug, say researchers writing in Arthritis & Rheumatology this month.
Retinopathy is the major adverse effect with hydroxychloroquine, which is the only medication that has shown improved survival in SLE. Reported risk factors include daily doses exceeding 400 mg, cumulative doses above 1000 g, use beyond 5 years, obesity, patients age greater than 60 years, and renal or hepatic dysfunction. Still, hydroxychloroquine, decreases disease flares, has anti-thrombotic and anti-diabetic effects, and is associated with complete renal remission when given in combination with mycophenolate mofetil. However, these benefits were shown using older dosing regimens.
In 2016, the American Academy of Ophthalmology (AAO) changed the recommended dosing of hydroxychloroquine from 6.5 mg/kg to less than 5 mg/kg, following a large retrospective study that found retinopathy rates of almost 20 percent by year 20 of use.
“It is not clear that the lower prescribed dose of hydroxychloroquine will have the same efficacy for SLE activity or the same protective role against cardiovascular risk factors and thrombosis. We addressed the frequency of hydroxychloroquine retinopathy and the role of hydroxychloroquine blood levels to identify those at greater future risk of retinopathy,” wrote the authors, led by Michelle Petri, M.D. M.P.H., of Johns Hopkins University School of Medicine Baltimore in Maryland.
This prospective cohort study included 537 patients (92 percent female) who underwent retinal testing, while hydroxychloroquine blood levels were obtained from 492 patients. There was 1 percent risk of retinopathy in the first five years, 1.8 percent from six to 10 years, 3.3 percent from 11-15 years, 11.5 percent from 16-20 years, and 8 percent after 21 years of use.
Among patients in the lowest mean tertile of hydroxychloroquine blood levels (0 to 741 ng/mL), the rate of retinopathy was 1.2 percent, 3.7 percent for those with blood levels of 741.5 to 1,176.5 ng/mL and 7.9 percent for patients with levels of 1,177 to 3,513 ng/mL (P for trend = 0.0027),
“Our data agree that the prevalence of hydroxychloroquine retinopathy, using newer screening technologies, is much higher than previously reported," the authors wrote.
The overall prevalence of hydroxychloroquine toxicity was 4.3 percent.
"Monitoring hydroxychloroquine blood levels is an important step to improved medication adherence in patients with SLE. We now introduce the concept of hydroxychloroquine blood level monitoring to reduce overdosage,” the authors wrote.
Higher toxicity rates were seen in patients with a high body mass index, patients older than 60 years (10.1 percent versus 0.5% for those aged <45) and with a longer duration of treatment. African Americans had lower daily doses of hydroxychloroquine than Caucasians (4.46 versus 4.84 mg), but there were no differences in blood levels or retinopathy rates between African Americans and Caucasians. The prevalence of retinal toxicity increased in accordance with the tertile blood levels, from 1.2 percent at the lowest to 6.7 percent at the highest tertile.
"The clinical impact of our study is that practitioners would be able to either decrease hydroxychloroquine dose or increase monitoring in patients with the highest tertile of blood levels," the authors wrote.
REFERENCE
Michelle Petri, Marwa Elkhalifa, Jessica Li, et al. “Hydroxychloroquine Blood Levels Predict Hydroxychloroquine Retinopathy.”Arthritis and Rheumatology. September 18, 2019. doi: 10.1002/art.41121