Focus on the Gut Microbiome to Manage Psoriasis
The gut microbiome has previously been considered a therapeutic target for the immune system, and new research suggests the microbiome’s composition can hold the key to managing psoriasis.
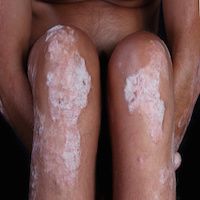
The gut microbiome has previously been considered a therapeutic target for the immune system, and new research suggests the microbiome’s composition can hold the key to managing psoriasis.
According to Herbert Dupont, MD, microbiome expert, “The intestinal bacteria look like a Persian carpet with tens of thousands of species of bacteria that form little interlocking walls to keep microbes that may cause mischief in the gut from producing — kind of walling them off. Well, when people get courses of antibiotics the diversity of these bacteria is depleted, and instead of a Persian carpet, it is a carpet with one or two colors in it, and now disease can occur.”
The research team used three different animal models to asses the interactions of the gut microbiome and specific immune system cells called TH17 that have previously been correlated to the formation of psoriatic lesions.
The team treated mice models with imiquimod (IMQ) — a potent immune activator cream used to induce psoriatic lesions in the animal. The mice were treated with antibiotics 2 weeks before psoriasis induction and continued until the end of the experiment.
And after measuring the gut microbiome, immune system, and degree of lesion formation, results showed:
· Germ free (GF) and conventional (CV) mice were more resistant to IMQ-induced skin inflammation than C57BL/6 mice
· The antibiotic treatment in CV mice caused significant changes in measures of gut microbiome diversity.
· The antibiotic treatment triggered an increase in certain gut bacteria like Lactobacillales and a major decrease in other bacteria like Coriobacteriales.
· IMQ induced a lower degree of local and systemic TH17 activation in both GF and CV mice.
What does this mean?
Consistent interactions between microbiota and the immune system are vital for maintaining host homeostasis, so any change in microbiota composition would cause a shift in immune system reactivity and trigger inflammatory diseases.
Researchers surmised the distinct interactions between the gut microbiome and the immune system could be a potential clinical target for therapeutic management and prevention of lesions in patients.