Lower Treatment Effectiveness for Secukinumab, Ustekinumab in Real-World Setting
There is a gap between the efficacy of biologics in trials and the real-world setting.
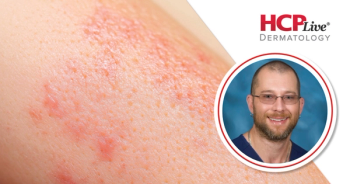
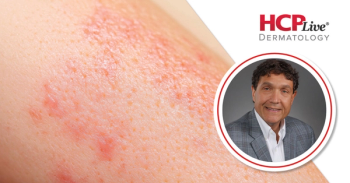
Secukinumab and ustekinumab have lower treatment effectiveness in a real-world clinical setting than in a trial setting, according to new study findings.
The findings suggested a gap between the efficacy of biologic therapies in an idealized trial setting and the effectiveness of biologic therapies in the real-world clinical setting in the treatment of
Zenas Yiu, PhD, and a team of investigators used data from BADBIR, a large registry of patients with moderate to severe psoriasis in the UK and Republic of Ireland who were received either a systemic therapy, a new oral small molecule systemic therapy, or a biologic therapy. The database contained detailed information on psoriasis characteristics, severity, and comorbid conditions. Data used by the team were from patients at least 18 years old with chronic plaque psoriasis. Patients needed to have at least 1 record of a Psoriasis Area and Severity Index (PASI) of at least 12 prior to the initiation of the investigated biologics.
The primary study outcome was the difference in the proportion of participants who achieved a PASI of 2 or lower after 12 months of therapy between the 2 comparator cohorts. Sensitivity analyses were performed using PASI 90 as an alternative secondary outcome.
To help with missing outcome data, the team used nonresponder imputation (NRI), last observation carried forward (LOCF), multiple imputation (MI), and inverse probability of censoring weighting (IPCW).
Overall, 1231 patients were included in the analysis, with 917 receiving ustekinumab and 314 receiving secukinumab. The PASI at 1 year was recorded for 42% of the secukinumab group and 45.5% of the ustekinumab group.
There were few serious adverse events reported within 12 months in both cohorts. In fact, fewer than 5 adverse events in each group were coded with the MedDRA high-level term of fungal infection disorders. The team noted no adverse events in the secukinumab cohort and 5 in the ustekinumab cohort were coded with the high-level term of noninfective colitis.
Yiu and the investigators found the proportion of participants who achieved a PASI of 2 or lower at 12 months was lower in their study compared with the CLEAR trial.
Every method achieved estimate agreement in the propensity score analysis except for NRI for risk difference (RD). On the other hand, complete case analysis (CCA) (RR, 1.55; 95% CI, 1.19-2.01), NRI (RR, 1.45; 95% CI, 1.04-2.03), IPCW (RR, 1.50; 95% CI, 1.14- 1.96), and MI (RR, 1.44; 95% CI, 1.07-1.94) all overestimated the RR in the propensity score-matched analysis, violating estimate agreement. The investigators found the propensity score-matched analysis performed better in estimation of RD, reaching estimate agreement for all missing outcome imputation methods (CCA: 19.5% [95% CI, .1-31.6]; NRI: 7.2% [95% CI, .4-13.9]; IPCW: 18.3% [95% CI, 5.5- 31.1]; LOCF: 14% [95% CI, 5.9-22.1]; MI: 16.9% [95% CI, 3.3-30.6]). LOCF with propensity matching (RR, .009; RD, -.003) resulted in the lowest standardized difference for RR and RD between Yiu’s study and the CLEAR trial.
In all analyses, secukinumab was superior to ustekinumab except under the NRI method.
Treatment with secukinumab resulted in a higher proportion of patients reaching a PASI of 2 or lower after 12 months of therapy compared with ustekinumab. Still, an efficacy-effectiveness gap was found for both treatments.
The study, “