Why Does This 63-Year-Old Male Diabetic Have a Shift in His Myloid Series?
This 63-year-old man with diabetes was hospitalized for bronchitis in a local community hospital and found to be anemic.
CLINICAL HISTORY
This 63-year-old man with diabetes was hospitalized for bronchitis in a local community hospital and found to be anemic.
The anemia was unresponsive to oral iron supplementation and B12 therapy and was not definitively characterized at that time. In the next few months, he required occasional transfusions. Bone marrow testing 3 months after his hospitalization revealed
erythroid hyperplasia and granuloma. A regimen of prednisone and a testosterone substitute was started 2 months later, with a
good clinical response.
Eight months later he was admitted to our university medical center with a chief complaint of progressive weakness and nonradiating, pleuritic chest pain. Bone marrow testing revealed a shift in the myeloid series, suggestive of an acute leukemia/chronic lymphocytic process.
PHYSICAL EXAMINATION
On physical examination he was afebrile, with mild scleral icterus; his chest was clear on percussion and auscultation, and his liver was slightly tender. Notable laboratory findings included positive stool guaiac (3+/4+); hematocrit, 14.3%; and hemoglobin, 4.6 g/dL.
A posterior anterior (PA) chest radiograph displayed a small density marginating the left upper lung bronchus, small nodules in both upper lungs, and mild blunting of the left
costophrenic sulcus (Figure 1).
During hospitalization, basilar rales were noted in both lungs, and an upper gastrointestinal series revealed two gastric ulcers.
Head and neck consultation revealed bilateral superficial ulcerations with secondary infection in the nasal lumen. Subsequent laboratory testing included cultures of sputum and bronchial brush specimens that grew Aspergillus species, and a right iliac bone biopsy that ruled out chronic lymphocytic leukemia.
RADIOGRAPHIC FINDINGS
PA and lateral chest radiographs (Figure 1) displayed multiple bilateral patchy densities, especially in the apices. Nodular densities were present between the posterior 4th and 5th ribs, and an amorphous density on the left between the posterior 5th and 6th ribs.
Findings in the right lung included several small nodules in the upper lobe and a radiolucency 1.5 cm in diameter at the level of the posterior 7th rib (Figure 2).
The lateral chest radiograph displayed a left upper lobe density overriding the proximal portion of the transverse thoracic aorta (Figure 3).
Pathology findings from a needle biopsy specimen of the left upper lung mass (Figure 4) displayed a coagulative necrosis with birefringent foreign particles and a fungus morphologically consistent with the genus Mucor. This confirmed the diagnosis of mu-
cormycosis, and the patient was treated with amphotericin B.
Discussion
Mucormycosis, a fungal infection (genus Mucor; class Zygomycetes), is a true opportunistic infection in that it cannot cause disease unless the host has an impaired immune system. It is usually acquired through inhalation and is most often present in patients with diabetes, leukemia or lymphoma, severe immunosuppression, or trauma mellitus; diabetes is the most common predisposing factor.
Patients on long-term corticosteroid therapy and transplant recipients (bone marrow and solid organs) are at risk. The mortality rate can be as high as 80%1or as low as 15%-30% in cases with early diagnosis and aggressive treatment.2
The incidence seems to be increasing slightly.3,4 Of interest, the infection was
found in a patient with severe wounds from the December 2004 tsunami in Southeast
Asia, suggesting that survivors of natural disasters are at increased risk.5
Clinical manifestations are generally grouped into 6 syndromes: rhinocerebral, pulmonary, cutaneous, gastrointestinal, central nervous system, and disseminated dis-
ease. Because the organism has an affinity for blood vessels, vascular invasion and tis-
sue necrosis are hallmarks of the disease.2
Mucormycosis localizes depending on the nature of the primary disease. In patients with diabetes, it usually occurs in ketoacidosis and often starts with paranasal sinus inflammation. A black mycelial mass may be detected in the nares.2
Inflammation can extend retro-orbitally and cause ophthalmoplegia. Necrosis of facial tissue is a dire outcome in late presentations.6 In diabetic patients, the lung is usually involved later in the disease after rhinocerebral manifestations.
Conversely, symptoms usually start in the lungs in the setting of leukemia, lymphoma, or long-termcorticosteroid therapy. Chest pain, fever, hemoptysis, and friction rub are common findings.
•The differential diagnosis should include fungal disease such as cryptococcosis, nocardiosis, aspergillosis, mucormycosis, coccidioidomycosis, and histoplasmosis and granulomatous diseases such as Wegener’s granulomatosis.
The diagnosis is confirmed by tissue biopsy (culture, histopathologic examination, or both), and imaging studies are needed to the ensure the diagnosis.
Chest radiographs, as in our patient, may display cavitation, air fluid levels, and solid mass-like densities.
In a patient with mucormycosis, however, magnetic resonance imaging (MRI) and computed tomography (CAT) scans usually display the destructive landmark anatomy in
advanced disease.
• Treatment should be aggressive and started as early as possible to limit the high mortality rate of mucormycosis. Management of the primary disease is essential, along with control of underlying immunosuppression and initial high-dose amphotericin B.1,2,4,7
Surgical debridement of necrotic tissue (which may include resection of lung tissue) is often necessary.4,6-8
Take-home messages from this case
Mucormycosis is a disorder that occurs only in patients with immunosuppression. Diabetes is the most common predisposing factor. Early recognition and treatment are crucial to limit the high mortality of advanced or untreated disease.
Rhinocerebral involvement (black eschars or discharge) is typically the initial manifestation in diabetic patients, with vascular invasion and tissue necrosis being the hallmarks of the disease.
Chest radiographs must be obtained for baseline comparison studies. It is not unusual that other symbiotic organisms such as E. coli and Candida albicans may be present.
Since other symbiotic organisms may be present, follow-up chest radiographs are necessary to document other organisms not growing while the patient is under going treatment
Treatment must be aggressive with amphotericin B, control of underlying disease, and often surgical debridement.
FIGURES
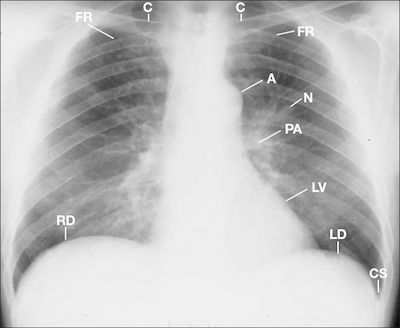
Figure 1 This posterior anterior chest radiograph displays a density marginating the left upper lung bronchus (N) and mild blunting of the left costophrenic sulcus (CS). A = aorta, C = clavicle , FR = first rib, LD = left hemidiaphragm, LV = left ventricle, PA = pulmonary artery; RD = right hemidiaphragm.
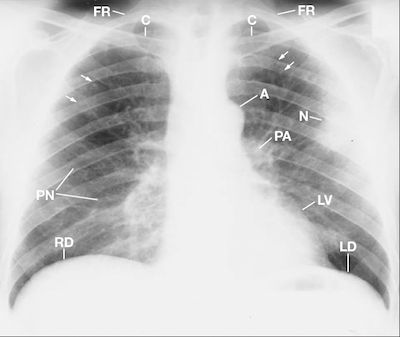
Figure 2 This posterior anterior chest radiograph displays an amorphous nodule (N) over the posterior left 5th intercostal space as the anterior 4th left rib crosses with nodules in the upper left lung (arrows). A = aorta, C=clavicle, FR = first rib, LD = left hemidiaphragm, LV = left ventricle, PA = pulmonary artery, PN = pneumatocele, RD = right hemidiaphragm.
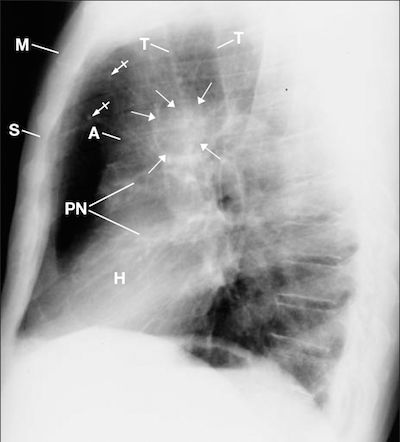
Figure 3 This lateral chest radiograph displays the left lung nodule (arrows) over the ascending aorta (A). Observe the small nodular densities within the anterior superior me-
diastinum (bar arrows). H = heart, M = manubrium, PN = pneumatocele, S = sternum, T = trachea
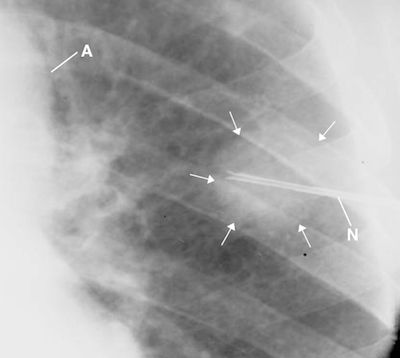
Figure 4 This fluoroscopic spot film documents the needle (N) biopsy of the left upper lung mass (arrows).9 Observe the lack of any densities marginating the mass. Mucormycosis was successfully cultured from the lung biopsy. A = aorta.
REFERENCES
1. Tedder M, Spratt JA, Anstadt MP, et al. Pulmonary mucormycosis: Results of medical and surgical therapy. Ann Thorac Surg 1994;57:1044-1040.
2. Jernigan JA. Mucormycosis (Zygomycosis, Phycomycosis). In: Hurst JW, ed. Medicine for the Practicing Physician. 4th ed. Stamford, Conn: Appleton & Lange; 2000:444-446.
3. Greenberg RN, Scott LJ, Vaughn HH, et al. Zygomycosis (mucormycosis): Emerging clinical importance and new treatments. Curr Opin Infect Dis 2004;17:517-525.
4. Pavie J, Lefaurie M, Lacroix C, et al. Successful treatment of pulmonary mucormycosis in an allogenic bone-marrow transplant recipient with combined medical and surgical therapy. Scand J Infect Dis 2004; 36:767-769.
5. Andresen D, Donaldson A, Choo L, et al. Multifocal cutaneous mucormycosis complicating polymicrobial wound infections in a tsunami survivor from Sri Lanka. Lancet 2005; 365:876-878.
6. Bhansali A, Bhadada S, Sharma A, et al. Presentation and outcome of rhino-orbital-cerebral mucormycosis in patients with diabetes. Postgrad Med J 2004;80:670-674.
7. Agarwal R, Kumar V, Gupta D. Pulmonary mucormycosis: Two of a kind. Eur J Intern Med 2006;17:63-65.
8. Karakurum B, Karatas M, Cagici AC, et al. Mucormycosis presenting with painful ophthalmoplegia. Acta Neurol Belg 2005;105:201-205.
9. Collins J D, Lung biopsies and patient care at university-affiliated hospitals: a 20-year experience. J Natl Med Assoc. 1990 Jul; 82(7): 485—491.
