Managing osteoarthritis: Current and future directions
ABSTRACT: Osteoarthritis (OA) was considered a disease of articularcartilage but now is thought to involve the entire joint. Research findingshave helped improve the understanding of disease progressionand the strategies used to alter the disease course. There is no laboratoryor pathological definition of OA; the diagnosis must be madeclinically. The approach to treatment should be tailored to specific patients.Topical agents often are used before oral medications. NSAIDsand cyclooxygenase-2 inhibitors remain the mainstay of therapy.Intra-articular corticosteroid injections have been shown to provideshort-term relief, and several hyaluronans are available for use inknee OA. Surgery may be required; modern implants provide significantpain relief and have proved to be durable and functional.(J Musculoskel Med. 2008;25:346-352)
ABSTRACT: Osteoarthritis (OA) was considered a disease of articular cartilage but now is thought to involve the entire joint. Research findings have helped improve the understanding of disease progression and the strategies used to alter the disease course. There is no laboratory or pathological definition of OA; the diagnosis must be made clinically. The approach to treatment should be tailored to specific patients. Topical agents often are used before oral medications. NSAIDs and cyclooxygenase-2 inhibitors remain the mainstay of therapy. Intra-articular corticosteroid injections have been shown to provide short-term relief, and several hyaluronans are available for use in knee OA. Surgery may be required; modern implants provide significant pain relief and have proved to be durable and functional. (J Musculoskel Med. 2008;25:346-352)
Of the estimated 46 million Americans who have doctor-diagnosed arthritis,1 at least 27 million are affected by clinically symptomatic osteoarthritis (OA).2 Although OA-the most common arthritis-is considered a disease of aging, it also frequently affects working adults, representing the largest cause of disability and lost work days.
OA traditionally was considered a disease of articular cartilage. Now it is thought to involve the entire joint. The diagnosis must be made clinically, because laboratory tests may not be helpful and radiographic findings do not necessarily correlate with symptoms. The goals of treatment are to palliate pain, maintain and improve function, and retard disease progression; the effort should be multidisciplinary and include patient education, physical therapy and occupational therapy, and nonpharmacological and pharmacological interventions.
In this article,we describe current approaches to the diagnosis and management of OA. We also take a look at potential future treatments.
CAUSES
OA represents the final common clinical pathway from a variety of disease processes. In the traditional view, it is considered primary if no triggering cause can be identified or secondary if a preceding source (eg, major trauma) has been noted. In either case, OA is thought to be largely mediated through aberrant biomechanical forces transmitted across the joints.
Risk factors for OA usually are separated into those associated with disease development (incident OA) or disease progression. The main factors associated with incident OA are aging, previous trauma, genetic predisposition, and obesity (Table 1). For disease progression, most studies have focused on the knees and hips; the chief factors include mechanical alignment, quadriceps strength, and intra-articular and bone marrow features.

Research has shown not only that OA involves all joint structures and is more than a degenerative disease of cartilage but also that biomechanics play a critical role in disease onset and progression.3,4 These findings (eg, persons with symptomatic medial knee OA overload their medial compartment,5 knee OA progression is associated with biomechanical loading,4 and knee loading is altered in association with pain severity6) have helped improve both the understanding of disease progression and the strategies used to alter the disease course.
DIAGNOSIS
Clinical pattern
Because there is no laboratory or pathological definition of OA, the diagnosis must be made clinically. The typical presenting symptom is pain that involves 1 or a few joints and starts during physical activity. In some cases, especially with interphalangeal (IP) joint involvement, patients may notice cosmetic changes, such as bony prominences, before experiencing pain (Table 2).

OA is not associated with systemic signs or symptoms of inflammation; therefore, there should be no significant or prolonged morning stiffness. Osteophytes, felt clinically as bony enlargements at the joint margins, are common; they may cause pain and even erythema of the overlying skin. Joint effusions are common but tend to be noninflammatory.
In advanced disease, involved joints may have limited range of motion because of severe articular or meniscal cartilage degeneration; intra-articular loose bodies and osteophyte growth also may be responsible. The most frequently involved joints include the distal IPs, proximal IPs, and first carpometacarpal of the hands; the knees; the hips; and the spine (Figure 1). The elbows, wrists, and ankles appear to be somewhat spared, except after major trauma or a sports injury.
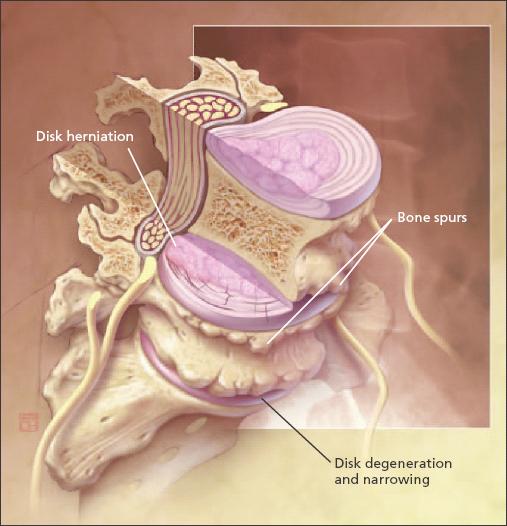
Figure 1 –
Problems associated with osteoarthritis (OA) in the lumbar spine include joint degeneration, bone spurs, and vertebral disk narrowing. The hands, knees, hips, and spine are the most frequently involved joints in OA.
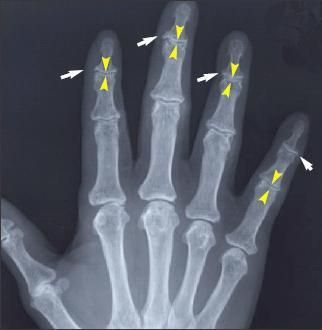
Figure 2 –
Osteoarthritis (OA) has characteristic radiographic features, but radiography typically is used to assess structural progression or to exclude other sources of pain. The distal interphalangeal (DIP), proximal interphalangeal (PIP), and first carpometacarpal are the joints most often affected in hand OA. Narrowed DIP and PIP (yellow arrows) joints and osteophytes (white arrows) are shown in this x-ray film.
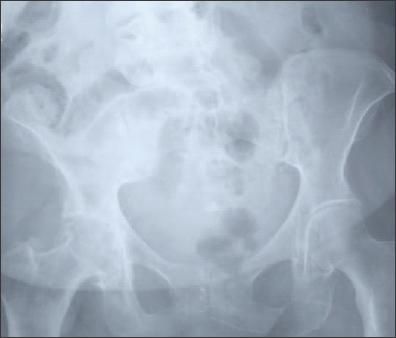
Figure 3 –
In patients with hip osteoarthritis, femoral or acetabular osteophytes and joint-space narrowing (superior, axial, and medial) may be seen on x-ray films.
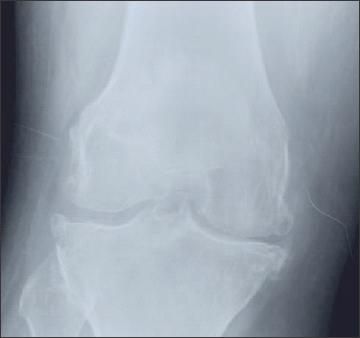
Figure 4 –
Asymmetrical joint-space narrowing, tibial and femoral osteophytes, and subchondral stenosis may be seen on x-ray films in patients with knee osteoarthritis.
Imaging
In practice, joint imaging generally provides adjunctive rather than diagnostic information. Although OA has characteristic radiographic features, radiography typically is used to assess structural progression or to exclude other sources of pain. In addition, the absence of radiographic findings characteristic of OA does not exclude the diagnosis, because cartilage is radiolucent and bony changes may not be present in early disease.
However, radiographic OA is defined by the presence of joint-space narrowing, which suggests loss of cartilage, osteophytes, and sclerosis of the subchondral bone (see Table 2; Figures 2, 3, and 4). Unlike in the inflammatory arthritides, the joint-space narrowing of OA is asymmetrical within the joint.
MRI is technologically ideal for imaging cartilage, and now advanced techniques can reliably and noninvasively quantify the glycosaminoglycan content and hydration of cartilage.7 However, there is little place for MRI in the routine diagnosis or management of OA.
Ultrasonography represents a noninvasive method of evaluating cartilage and meniscus integrity and has utility for guiding arthrocentesis. Although this technique is not yet popular for evaluation and management of clinical OA in the United States, it has become accepted in Europe.8
Laboratory findings
Because no routine laboratory tests are useful for the diagnosis or clinical management of OA, great effort has been expended in the search for biomarkers that might help. In principle, they might be derived from serum, synovial fluid, or urine.
A large variety of precise markers of cartilage and bone metabolism and of inflammation have been evaluated longitudinally, alone and in combination, and several have shown reasonable correlation with disease progression in large OA patient populations.9 Currently, however, no biomarkers are useful in the clinical care of individual OA patients, at least partly because although OA is not generally considered a systemic disease, there is a great deal of subclinical disease burden. Therefore, joints undergo biological degenerative processes for years (or decades) before they become symptomatic; all biologically involved joints,whether symptomatic or not,would influence biomarker levels, falsely elevating the measured levels.
TREATMENT
The approach to treatment should be tailored to specific patients because OA is heterogeneous, involves different joints, and ranges in severity from mild to severe. However,the underlying principles of managing OA-improving pain and function and retarding disease progression-have not changed for decades.
Strategies for relieving pain include adjunctive, nonpharmacological approaches as well as medications. Although the use of analgesics and NSAIDs is widespread and important, the adjunctive modalities that have been shown to provide pain relief should not be neglected (Table 3).

Nonpharmacological strategies
Patient education and support. Self-help programs alone can improve outcomes in OA.10 Patients should be provided thorough information about the natural course of OA, their role in disease management, and appropriate expectations. Many patients are comfortable using Internet resources, and a variety of Web sites provide useful information about OA aimed at a lay audience.They include the following: the Arthritis Foundation (www.arthritis.org/osteoarthritis-disease-focus.php), the NIH (www.niams.nih.gov/Health_Info/Osteoarthritis), and the American College of Rheumatology (www.rheumatology.org/public/factsheets).
Physical therapy. Patients should be instructed and trained in appropriate nontraumatic exercise regimens to improve range of motion. Adjunctive therapeutic modalities, such as diathermy (therapeutic heat) and ultrasonography, may be discussed.
Occupational therapy. This is used frequently in patients who have inflammatory arthritis to provide implements that assist in activities of daily living; it is equally useful in OA. An occupational therapist can evaluate the patient's functional ability, provide assistive devices as needed, and teach joint protection techniques and energy conservation skills.
Weight loss. Obesity is a major risk factor for OA. Weight loss, when practical, may result in improved function as well as pain palliation.11
Ice and diathermy. For theoretical reasons, ice may be expected to help reduce the local inflammatory component associated with OA flares. Similarly, many patients think that they obtain benefit from therapeutic heat, although systematic evidence is sparse.
Exercise. Exercise and strengthening regimens provide substantial pain relief in OA, and patients should be actively encouraged to adopt a regular strengthening program.12 As with weight loss, long-term patient compliance is poor, but exercise clearly is an effective strategy for motivated persons.
Complementary and alternative medicine (CAM) and nutritional supplements. The overwhelming majority of patients with arthritis have tried a CAM modality to relieve their pain.13 In one study, at least half of patients with knee OA had used at least 1 CAM modality in the prior 20 weeks.14 Various studies have reported some benefit with the use of many modalities, including acupuncture and some herbal supplements, although the effect sizes tend to be low and concern remains about systematic bias in these reports.
The most discussed supplements are glucosamine and chondroitin sulfate, which remain popular even after the well-powered NIH-sponsored Glucosamine/Chondroitin Arthritis Intervention Trial (GAIT) reported no significant benefit from these agents at 2 years when used individually or in combination.15 Controversy remains about the preparations that were used in GAIT and whether specific subgroups may have had significant benefit. Regardless, a large percentage of patients with OA continue to use these agents and perceive that they are beneficial. If the preparations are manufactured with adequate quality control, the adverseeffect profile should be benign.
Pharmacological therapy
Because the main goal of pharmacological therapy is to provide the most effective pain relief with the least toxicity, topical agents often are used before oral medications (Table 4). Capsaicin cream, approved in the United States for managing OA pain, is thought to act locally by depleting the neurotransmitter substance P; it must be applied long term to achieve a response. Topical NSAIDs have been available for OA in Europe and Canada for some time; in the United States, a topical diclofenac preparation is FDA-approved for use in OA.

When the use of topical agents is impractical or the results are insufficient, pure analgesics usually are tried. Acetaminophen is the first systemic pharmacological therapy recommended for hip or knee OA by the American College of Rheumatology, European League Against Rheumatism, American Academy of Orthopaedic Surgeons, and other organizations.16
An influential early study suggested that acetaminophen may be therapeutically equivalent to NSAIDs at 4 weeks.17 However, the bulk of the literature comparing acetaminophen with placebo and with NSAIDs over longer periods suggests that acetaminophen is inferior to NSAIDs and may not be clinically superior to placebo for longer term relief of OA pain.18,19
Other pure analgesics that have been shown to be effective are tramadol, a centrally acting analgesic, and opioid analgesics. However, both have a high incidence of intolerable adverse effects.
Despite safety concerns and recent publicity about cardiovascular risks, NSAIDs and cyclooxygenase (COX)-2 inhibitors remain the mainstay of OA therapy (Table 5); they are the only agents that consistently have been shown to retain pain palliation effects in OA over long periods. In GAIT, for example, although neither glucosamine nor chondroitin sulfate performed significantly better than placebo, the COX-2 comparator provided substantially better pain relief at 2 years.15
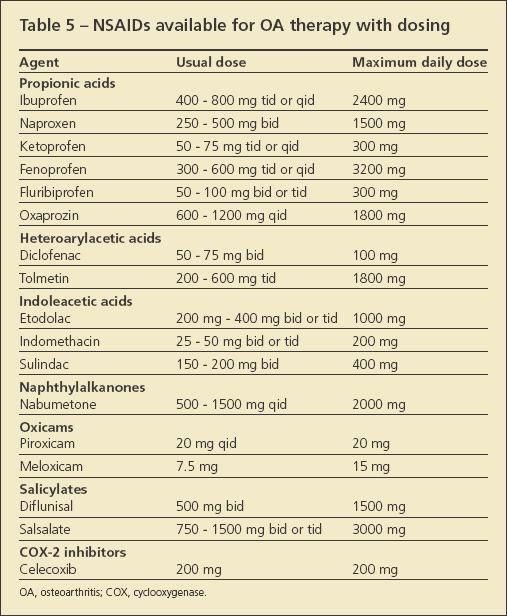
The choice of which NSAID to use should be based on several factors, such as dosing convenience, patient's and physician's comfort, and price. When NSAIDs are used in patients who are at increased risk for adverse GI events, adding either a proton pump inhibitor or misoprostol generally is recommended. Patients who are taking NSAIDs should be monitored for renal toxicity, especially if they are older, are hypertensive, or have diabetes mellitus.
Intra-articular therapy
Corticosteroids. Intra-articular corticosteroid injections have been shown to provide short-term relief, especially for painful flares; they also are useful for relieving pain that is unresponsive to optimal systemic therapy. Intra-articular injections in the same joint more than 3 or 4 times a year has been discouraged because of concerns about potential adverse effects to the articular cartilage and the surrounding joint structures.
Hyaluronans. Intra-articular injection of the hyaluronans is referred to as viscosupplementation because originally it was intended to increase the viscosity of the synovial fluid in OA to provide more normal tribological properties. However, because the half-life of the hyaluronan in vivo is quite short, the pain-palliating effects probably are the result of non–viscosity-related mechanisms.
Several hyaluronans are available for use in knee OA. Each has been shown to provide at least some pain palliation that often lasts for 6 months or longer. Although controversy remains about the magnitude of pain relief that actually results from using these agents, they are somewhat safe and well-tolerated.
Surgery
Patients whose symptoms are not controlled adequately with medical therapy and who have moderate to severe pain and functional impairment should be considered for surgery, especially if the knees or hips are the symptomatic joints. Modern implants provide significant pain relief and have proven to be durable and functional.
Arthroplasty has markedly improved the quality of life of patients with knee or hip OA and represents one of the most significant advances in OA management over the past 30 years.20 In addition to arthroplasty, the variety of procedures used for OA include arthroscopic debridement, arthrodesis, and joint realignment techniques. Several other approaches to OA therapy are under investigation by the pharmaceutical and biotechnology industries and by academic researchers (see the Box, "Osteoarthritis therapies on the horizon," below).
Osteoarthritis therapies on the horizon
New approaches to osteoarthritis (OA) therapy currently being investigated include attempts to identify disease-modifying OA drugs (DMOADs), tissue engineering for reconstituting intact cartilage and joint tissues, and biomechanically active methods for reducing aberrant loading in OA-involved joints. Likely targets for DMOAD therapy include each step in the degradative pathway of cartilage and in the remodeling of OA bone; for example, a variety of inhibitors of the aggrecanases and other proteases involved in the early degradation of cartilage matrix are in various stages of development.
Attempts to develop biomechanically functional cartilage constructs using various tissue engineering technologies, mesenchymal stem cells, or autologous chondrocytes have received a great deal of attention becauseof the application of these techniques to isolated chondral defects21 and because mesenchymally derived tissues appear to represent a particularly good target for stem cell technology. However, any long-term solution to modifying OA progression probably will need to incorporate biomechanically active strategies, because replacing degraded joint structures with functional constructs alone will not prevent rapid disintegration in the presence of ongoing aberrant biomechanical forces.
A variety of biomechanical strategies are under investigation. They include attempts to simulate the unloading effects of barefoot walking through shoe modification, 22 altering medial knee loads through the use of wedged orthotic inserts in shoes, and the wearing of unloading braces at the knee.
References:
References1. Centers for Disease Control and Prevention (CDC). Prevalence of doctor-diagnosed arthritis and arthritis-attributable activity limitation-United States, 2003-2005 [published correction appears in MMWR Morb Mortal Wkly Rep. 2006;55:1129]. MMWR Morb Mortal Wkly Rep. 2006;55:1089-1092.
2. Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States, part II. Arthritis Rheum. 2008;58:26-35.
3. Hurwitz DE, Sumner DR, Block JA. Bone density, dynamic joint loading and joint degeneration: a review. Cells Tissues Organs. 2001;169: 201-209.
4. Shakoor N, Hurwitz DE, Block JA, et al. Asymmetric knee loading in advanced unilateral hip osteoarthritis. Arthritis Rheum. 2003;48: 1556-1561.
5. Baliunas AJ, Hurwitz DE, Ryals AB, et al. Increased knee joint loads during walking are present in subjects with knee osteoarthritis. Osteoarthritis Cartilage. 2002;10:573-579.
6. Hurwitz DE, Ryals AR, Block JA, et al. Knee pain and joint loading in subjects with osteoarthritis of the knee. J Orthop Res. 2000;18: 572-579.
7. Gray ML, Burstein D, Kim YJ, Maroudas A. 2007 Elizabeth Winston Lanier Award Winner: magnetic resonance imaging of cartilage glycosaminoglycan: basic principles, imaging technique, and clinical applications. J Orthop Res. 2008;26:281-291.
8. Grassi W, Filippucci E, Farina A. Ultrasonography in osteoarthritis. Semin Arthritis Rheum. 2005;34(6 Suppl 2):19-23.
9. Rousseau JC, Delmas PD. Biological markers in osteoarthritis. Nat Clin Pract Rheumatol. 2007;3:346-356.
10. Devos-Comby L, Cronan T, Roesch SC. Do exercise and self-management interventions benefit patients with osteoarthritis of the knee? A metaanalytic review. J Rheumatol. 2006;33:744-756.
11. Christensen R, Astrup A, Bliddal H. Weight loss: the treatment of choice for knee osteoarthritis? A randomized trial. Osteoarthritis Cartilage. 2005;13:20-27.
12. Fransen M, McConnell S, Bell M. Exercise for osteoarthritis of the hip or knee. Cochrane Database Syst Rev. 2003;(3):CD004286.
13. Quandt SA, Chen H, Grzywacz JG, et al.Use of complementary and alternative medicine by persons with arthritis: results of the National Health Interview Survey. Arthritis Rheum. 2005; 53:748-755.
14. Ramsey SD, Spencer AC, Topolski TD, et al. Use of alternative therapies by older adults with osteoarthritis. Arthritis Rheum. 2001;45:222-227.
15. Clegg DO, Reda DJ, Harris CL, et al. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med. 2006;354:795-808.
16. Poitras S, Avouac J, Rossignol M, et al. A critical appraisal of guidelines for the management of knee osteoarthritis using Appraisal of Guidelines Research and Evaluation criteria. Arthritis Res Ther. 2007;9:R126.
17. Bradley JD, Brandt KD, Katz BP, et al. Comparison of an antiinflammatory dose of ibuprofen, an analgesic dose of ibuprofen, and acetaminophen in the treatment of patients with osteoarthritis of the knee. N Engl J Med. 1991;325:87-91.
18. Case JP, Baliunas AJ, Block JA. Lack of efficacy of acetaminophen in treating symptomatic knee osteoarthritis: a randomized, doubleblind, placebo-controlled comparison trial with diclofenac sodium. Arch Intern Med. 2003;163:169-178.
19. Towheed TE, Judd MJ, Hochberg MC, Wells G. Acetaminophen for osteoarthritis. Cochrane Database Syst Rev. 2003;(2):CD004257. [Update in Cochrane Database Syst Rev. 2006;(1):CD004257.]
20. Crawford RW, Murray DW. Total hip replacement: indications for surgery and risk factors for failure. Ann Rheum Dis. 1997;56:455- 457.
21. Brittberg M, Lindahl A, Nilsson A, et al. Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N Engl J Med. 1994;331:889-895.
22. Shakoor N, Block JA. Walking barefoot decreases loading on the lower extremity joints in knee osteoarthritis. Arthritis Rheum. 2006;54:2923-2927.