Psoriasis Patients with Arthralgia At Increased Risk of Psoriatic Arthritis Development
In this analysis, investigators sought to assess PsA development probability in subclinical PsA as well as to characterize symptoms and patterns of subclinical PsA.
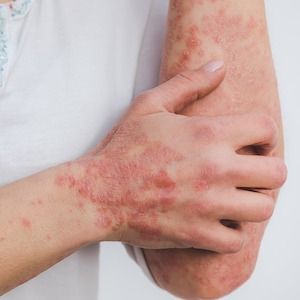
Individuals with psoriasis with unexplained arthralgia and non-inflammatory symptoms are at a far greater risk of psoriatic arthritis (PsA) development, according to new findings, providing new information on subclinical PsA.1
These findings resulted from new research conducted due to the lack of real-world data on subclinical PsA, which is described as the existence of arthralgia in psoriasis patients. This was considered invaluable given the noted higher risk of PsA among those with subclinical PsA.
The new analysis was led by Alen Zabotti, from the department of medical and biological sciences at the University Hospital Santa Maria della Misericordia Rheumatology Clinic in Udine, Italy. Zabotti et al. acknowledged that a prior systematic literature review had indicated the lack of data on the transition between psoriasis to PsA.2
“These could help to better define outcomes for PsA prevention studies in the context of prevention/interception of PsA and early diagnosis,” Zabotti and colleagues wrote. “Therefore, this study investigated the subclinical stage of PsA in terms of risk of progression and clinical symptoms, and to describe the clinical features of new-onset PsA in (psoriasis) cases.”3
Background and Methods
The investigators assessed data drawn from 2 ongoing European prospective cohorts of individuals with psoriasis, looking at both the Italian cohort, RAPSODI (Ultrasonographic Risk Factors to Develop Psoriatic Arthritis in Psoriatic Patients with and Without ClinIcal Arthralgia), and the German PACE cohort (Psoriasis and Psoriatic Arthritis Cohort Erlangen), both of which were longitudinal studies.1
Subjects featured within the 2 cohorts had been given a diangosis with definite psoriasis of the skin or nails by dermatologists. They were also enriched for PsA risk, with many having arthralgia suggesting a potential risk of PsA development.
The investigators looked at study participants that did not have clinical PsA by the time of their entry to the cohort and those with no visible swelling of joints. Subjects with a history of enthesitis, synovitis, dactylitis, or inflammatory back pain were not included in the research, and those who were included were reassessed every half year, being told by the team to report inflammatory symptoms to their designated rheumatologists.
In the period between January 2017 - December 2022, there had been 215 Italian participants enrolled in RAPSODI with psoriasis, of which 66.0% were shown to have baseline arthralgia indicating the presence of subclinical PsA. Within those in the PACE cohort, 169 participants in total between January 2011 - July 2018, all were determined to have subclinical PsA.
The investigators diagnosed new-onset clinical PsA at the time of follow-up through the use of experienced rheumatologists and with CASPAR criteria fulfillment. The team would also characterize the manifestations of subjects’ new-onset clinical PsA by enthesitis, dactylitis, peripheral arthritis, and axial involvement.
Additionally, the team recorded participants’ clinical features at the time of PsA diagnosis, some of which included enthesitis index, joint counts, and presence of dactylitis.
Findings
There were 384 individuals given a diagnosis of psoriasis who took part in the study, as well as an average follow-up duration of 33.0 months. Additionally, 80.9% of these individuals were shown to have subclinical PsA, indicating there had been an incidence rate of PsA of 7.7 per 100 patient-years.
The investigators reported that their subclinical PsA patients had a significantly higher risk of developing PsA as opposed to subjects that had psoriasis exclusively (HR=11.7, 95% CI 1.57 to 86.7, P = .016). The research team noted that the cumulative incidence function (CIF) estimated the probability of new-onset PsA was about 9.4% (95% CI 4.7% - 10.6%) by the 12-month mark and that it was 22.7% (95% CI 17.2% - 28.6%) by the 36-month mark.
A notable finding was that 58.9% of subjects had been shown to have inflammatory symptoms during the months immediately preceding their diagnosis of PsA, but 83.9% were found to have non-inflammatory symptoms before their PsA diagnosis. Additionally, the most frequent pattern of presentation was shown to be peripheral joint swelling for 82.1% of those with PsA.
“We have confirmed that patients with (psoriasis) experiencing unexplained arthralgia, also reporting non-inflammatory symptoms, are at a significantly elevated risk of developing PsA,” they wrote. “Additionally, peripheral arthritis, mainly oligoarthritis, is the most common PsA presentation in cohorts with PsO enriched for arthralgia.”
References
- Zabotti A, Fagni F, Gossec L, et alRisk of developing psoriatic arthritis in psoriasis cohorts with arthralgia: exploring the subclinical psoriatic arthritis stageRMD Open 2024;10:e004314. doi: 10.1136/rmdopen-2024-004314.
- De Marco G , Zabotti A , Baraliakos X , et al . Characterisation of Prodromal and very early Psoriatic arthritis: a systematic literature review informing a EULAR Taskforce. RMD Open 2023;9:e003143. doi:10.1136/rmdopen-2023-003143.
- Eder L , Lee K-A , Chandran V , et al . Derivation of a multivariable Psoriatic arthritis risk estimation tool (PRESTO): a step towards prevention. Arthritis Rheumatol 2023. doi:10.1002/art.42661.