A Woman with Progressive Cough, Weakness, and Weight Loss
As any pulmonary disease can mimic sarcoidosis, its diagnosis must be supported by all clinical methods involving follow-up radiographs and biopsy.

James D. Collins, MD
This 40-year-old female nurse presented with progressive weakness, cough, and wheezing. Though her sputum cultures, skin tests, and bone marrow biopsies were negative, her pulmonary function tests (PFTs) revealed mild decreased vascular flow and vital capacity, and her liver biopsy was positive for non-caseating granulomas with giant cells consistent with sarcoidosis.
After the patient was treated with prednisone and anti-tuberculosis (TB) therapy, she was discharged. However, she continued to have night sweats and coughing episodes that produced white and yellow sputum. She also lost 25 lbs over 3 months and noticed enlarged, supraclavicular lymph nodes. Her subsequent lymph node biopsy and percutaneous lung biopsy both revealed granulomatous inflammation consistent with TB.
Radiographic Findings
This woman’s posterior-anterior (PA) (Figure 1, 2) and lateral (Figure 3) plain chest radiographs displayed bilateral rounding of the shoulders, obesity, forward shift of the cervicothoracic spine, bilateral hilar lymph nodes, and peritracheal adenopathy, particularly on the right. An apical density was displayed on the right and a nodular, non-calcified density was present in the left mid-lung field.
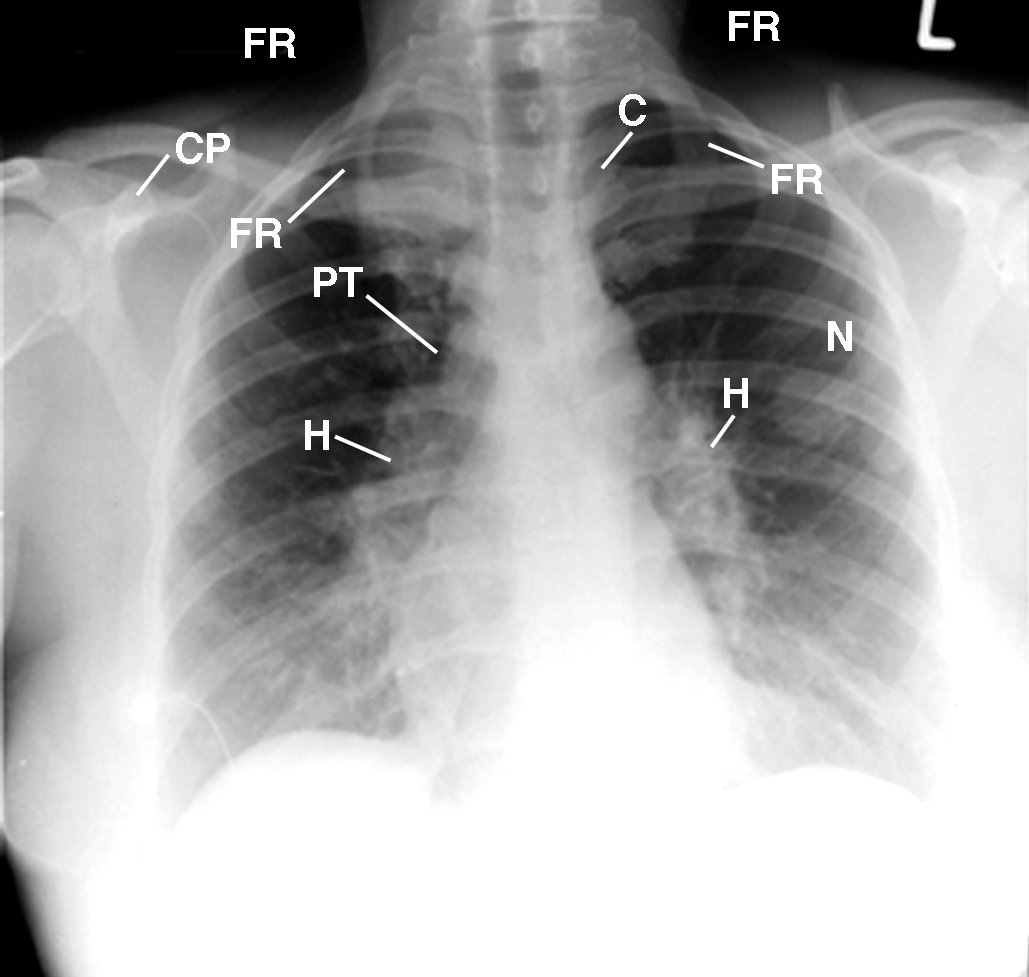
Figure 1 This PA chest radiograph displays the patient leaning to the right, marked increased slope of the first ribs (FR), forward-rotated heads of the clavicles (C) over the asymmetric posterior fourth ribs, a drooping right shoulder as compared to the elevated left shoulder, decreased density of the left supraclavicular fossa (not labeled) as compared to the right, cardiomediastinal structures displaced down and to the right of the midline, wire lead (not labeled) over the lateral region of the anterior segment of the right lower lobe (M), right peritracheal (PT) and bilateral hilar lymph nodes (H), and peritracheal adenopathy (PT), particularly on the right. An apical density (not labeled) is present on the right with a 2 1/2 x 3.0 cm nodular, non-calcified density (not labeled) in the left mid-lung field. CP= coracoid process; L= left.
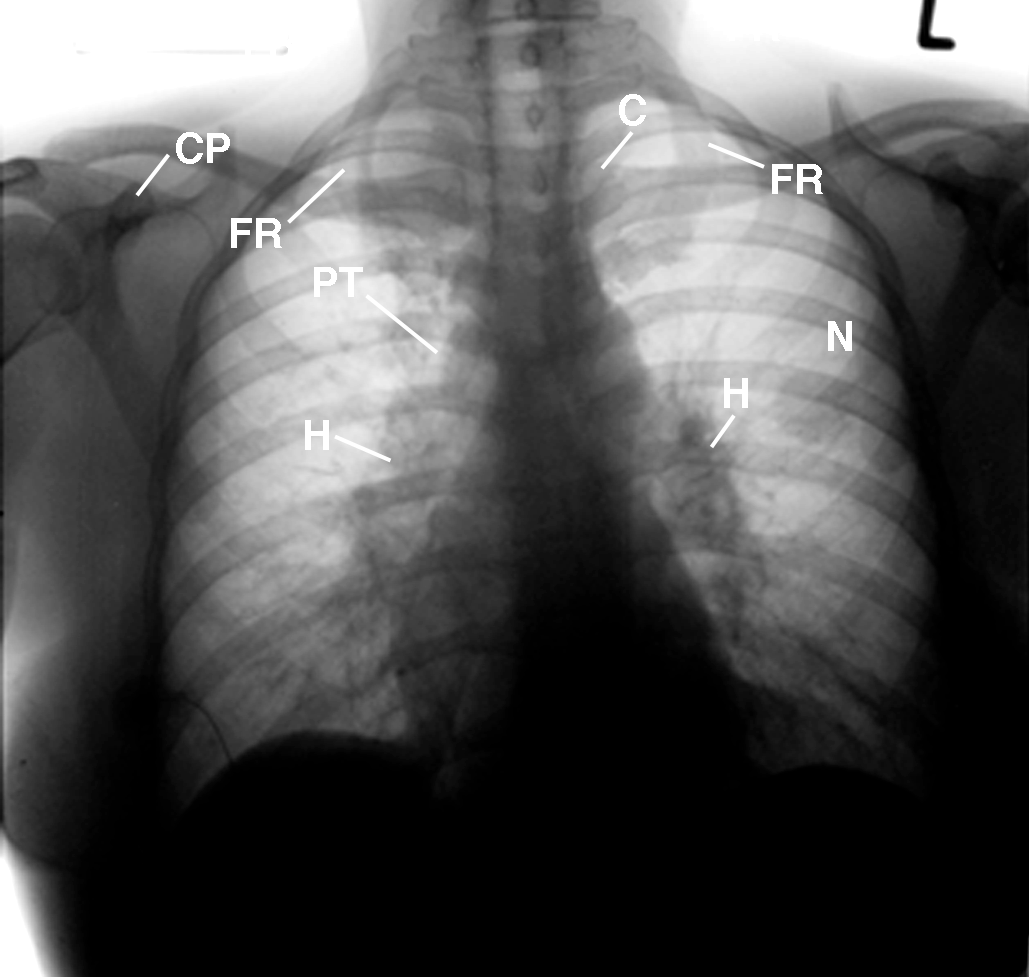
This inverted image of Figure 1 provides a different perspective of the landmark anatomy of the soft tissues of the thorax, chest wall, and osseous structures.
Figure 2
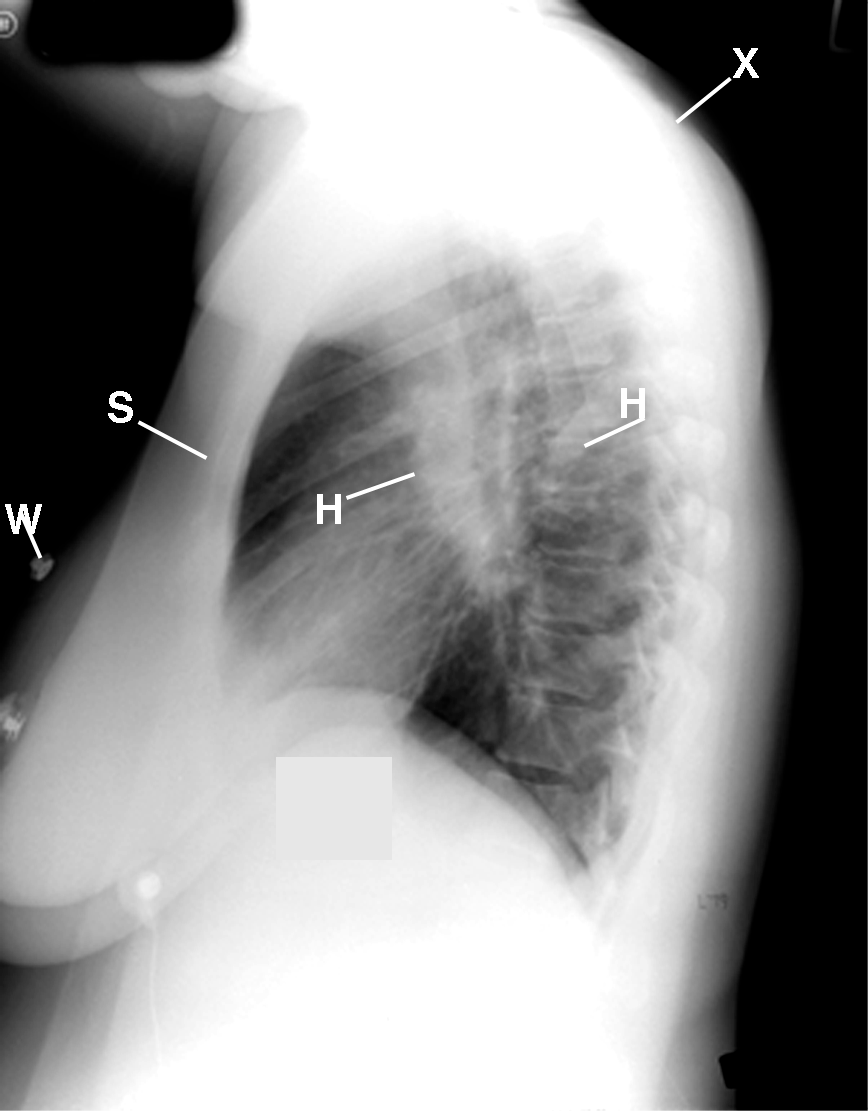
Figure 3 This lateral chest radiograph displays the density of the large hilar lymph nodes. Observe the moderate fat deposition over the neck, chest walls, and abdomen (not labeled) and the forward posture of the rounding of the shoulders (X). S= sternum, W= wire lead.
Diagnosis
Diagnostic considerations for this patient included TB, fungal disease, lymphoma, carcinoma, pneumoconiosis, HIV infection, and sarcoidosis. Ultimately, the woman was diagnosed with sarcoidosis, based on the findings from her chest radiographs and the non-caseating granulomas displayed in her lung, liver, and lymph node biopsies.1-2
Discussion
Sarcoidosis is an inflammation in which lymphocytes become overactive. These fervid white blood cells release chemical substances, which cause a collection of inflammatory cells known as granulomas to crop up in various organs of the body.
Although sarcoidosis is a multisystem disorder, it affects the lungs 90% of the time; thus, it is primarily classified as a pulmonary disease. While there may not be any symptoms of lung involvement at all, chest pain, cough, and chest tightness are the most common complaints of pulmonary sarcoidosis. Regardless of whether symptoms are present, a sarcoidosis patient’s chest X-ray will almost always appear abnormal.
Common radiographic features include bilateral hilar lymphadenopathy, interstitial changes, and enlarged right paratracheal lymph nodes similar to the “pawn shop sign” displayed in this presentation. Large, bullous areas and spontaneous pneumothorax may also occur, while severe scarring known as fibrosis of the lungs can cause respiratory failure.
Take-Home Message
The many radiographic findings in pulmonary sarcoidosis make an accurate diagnosis difficult. As any pulmonary disease can mimic sarcoidosis — including bronchial carcinoma, TB, lymphoma, chronic lymphatic leukemia, and fibrosis masquerading as asbestosis3-4 — its diagnosis must be supported by all clinical methods involving follow-up radiographs and biopsy. For fascial plane landmark anatomy, magnetic resonance imaging (MRI) is suggested over computed tomography (CT). Family physicians must remember that lymph node margins are usually more sharply defined in sarcoidosis than infections.4
References
1. Harrion TR, Wintrobe MM. (1974). Harrison’s Principles of Internal Medicine. (7th ed.). McGraw-Hill.
2. Mitchell DN, Scadding JG. Sarcoidosis. American Review of Respiratory Diseases. 1974;110:774-802.
3. Batra P, Gomes A, Collins JD. Sarcoid lymphadenopathy: an unusual cause of ventilation-perfusion quotient mismatch on lung scanning. J Natl Med Assoc. 1985 Nov;77(11):938-9, 943.
4. Collins JD. A young African-American male with dyspnea on exertion and cough. Family Practice Recertification. 2013 Dec;31(3). http://www.hcplive.com/publications/family-practice-recertification/2013/December2013/A-Young-African-American-Male-with-Dyspnea-on-Exertion-and-Cough.
About the Author
James D. Collins, MD, is Professor and General Radiologist in the Department of Radiology at the UCLA David Geffen School Medicine. He formerly served as Director of the required medical student training for the department and President of the James T. Case Radiologic Foundation. Collins has an extensive background of publications, consultations, and editorial positions, including his current post as the Radiology Editor for the Journal of the National Medical Association. He specializes in bilateral 3-dimensional MRI and MRA imaging of the brachial plexus and has been performing those studies since 1985.
