Chest Radiograph Cost-effectively Detects Benign Pulmonary Lesion
Hamartomas can be found incidentally via chest radiograph, a cost-effective screening tool that should always be part of the initial evaluation in patient care.

James D. Collins, MD
A 20-year-old female patient presented with complaints of severe headaches. Her remarkable physical examination and laboratory findings included a blood pressure of 250/150 and a serum creatinine concentration of 3.5 mg/dL. Contracted kidneys were discovered on an intravenous pyelography, and after further evaluation, a diagnosis of chronic glomerulonephritis was made.
A solitary nodule was detected on a plain chest radiograph that was found incidentally during the patient’s workup, though she had neither respiratory symptomatology nor any history to suggest previous pulmonary disease. Poster-anterior (PA) and lateral chest radiographs were then obtained.
Radiographic Findings
This woman’s PA chest radiograph (Figure 1) displayed a sharply demarcated nodule in the region of the anterior segment of her left upper lung, although the lateral view (Figure 2) implied the nodule could fall within the superior segment of the left lower lobe, which would be significant for a surgical approach. Faint calcifications were suggested within the nodule.
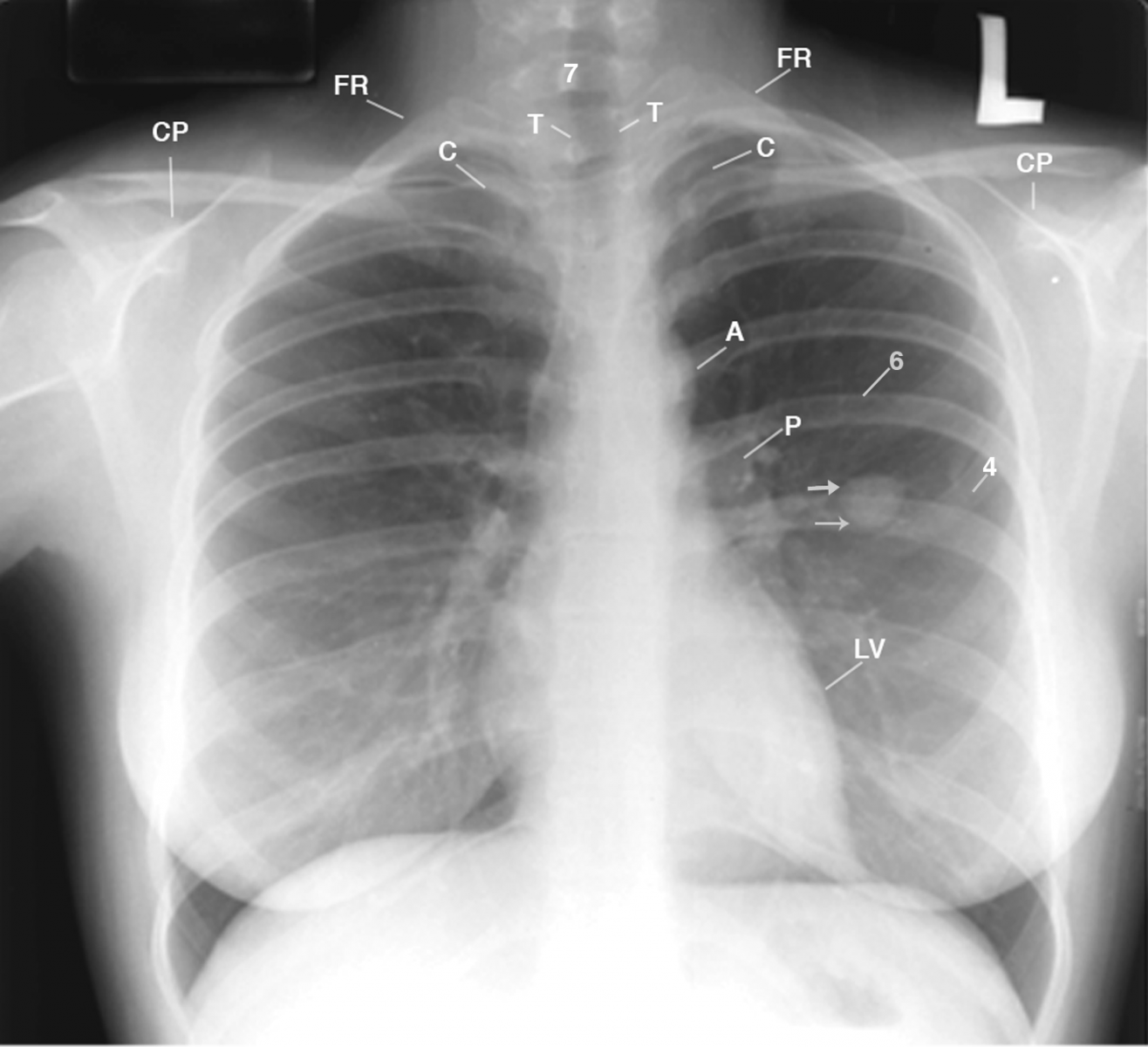
Figure 1
This PA chest radiograph displays the patient leaning right, accentuating the right concave scoliosis of the cervicothoracic spine (not labeled); a drooping right shoulder, as compared to the left shoulder; the anterior rotated head of the right clavicle (C) over the posterior second right intercostal space, as compared to the head of the left clavicle over the high left first rib (FR); amorphous density medial to the left anterior fourth rib (4) crossed by posterior left sixth rib (6); and small densities within a sharply marginated left pulmonary nodule (3 arrows). A= aorta; CP= coracoid process; P= pulmonary artery; LV= left ventricle; T= trachea.
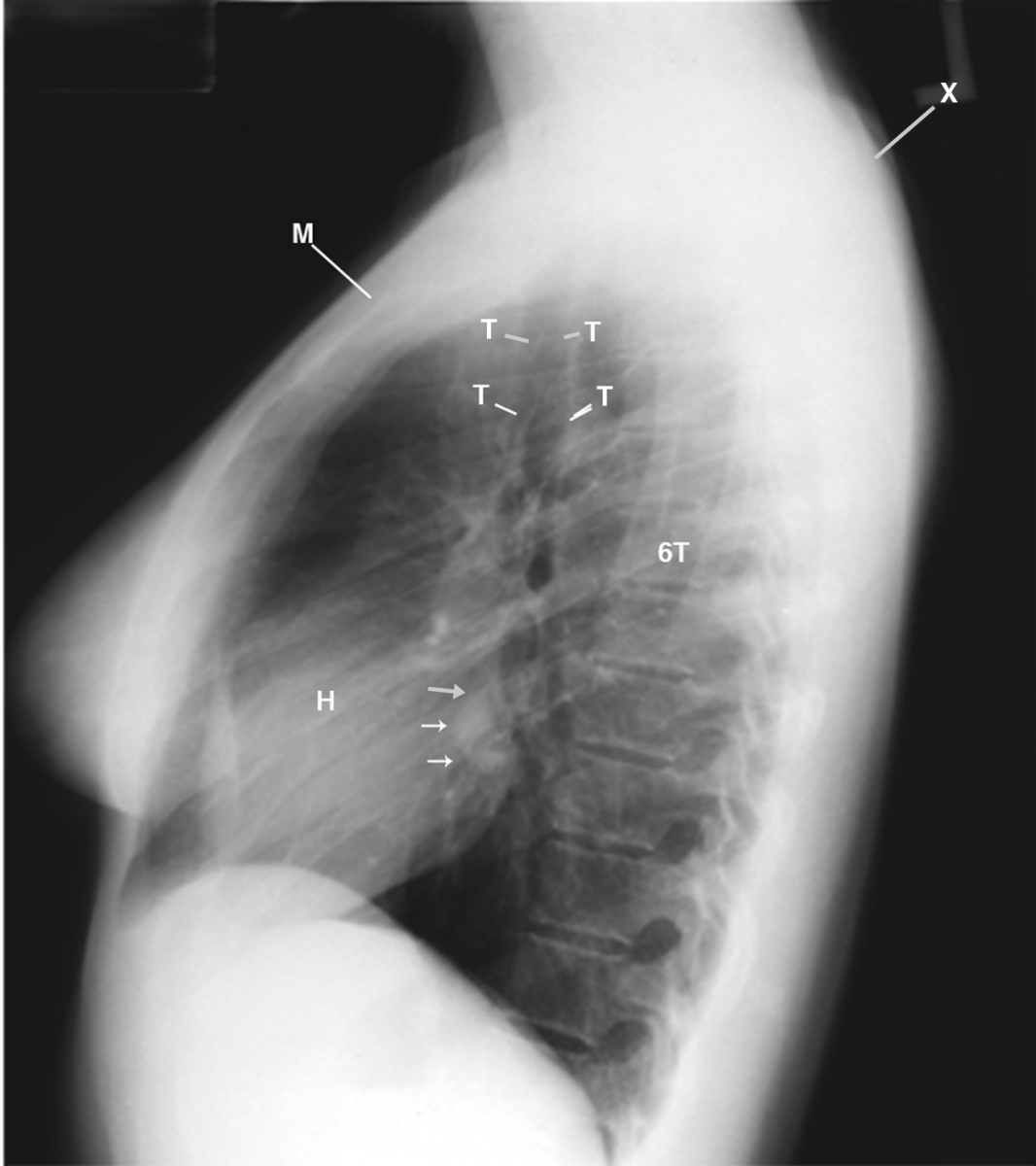
This lateral chest radiograph displays the solitary left lung nodule (arrows); rounding of the shoulders (X), and backward manubrium (M) that places the heads of the clavicles (not labeled) in close proximity to the first ribs (not labeled). H= heart; 6T= sixth thoracic vertebra.
Figure 2
An enlarged linear tomogram (Figure 3) identified distinct calcifications in a “popcorn” configuration that is pathognomonic of hamartoma.
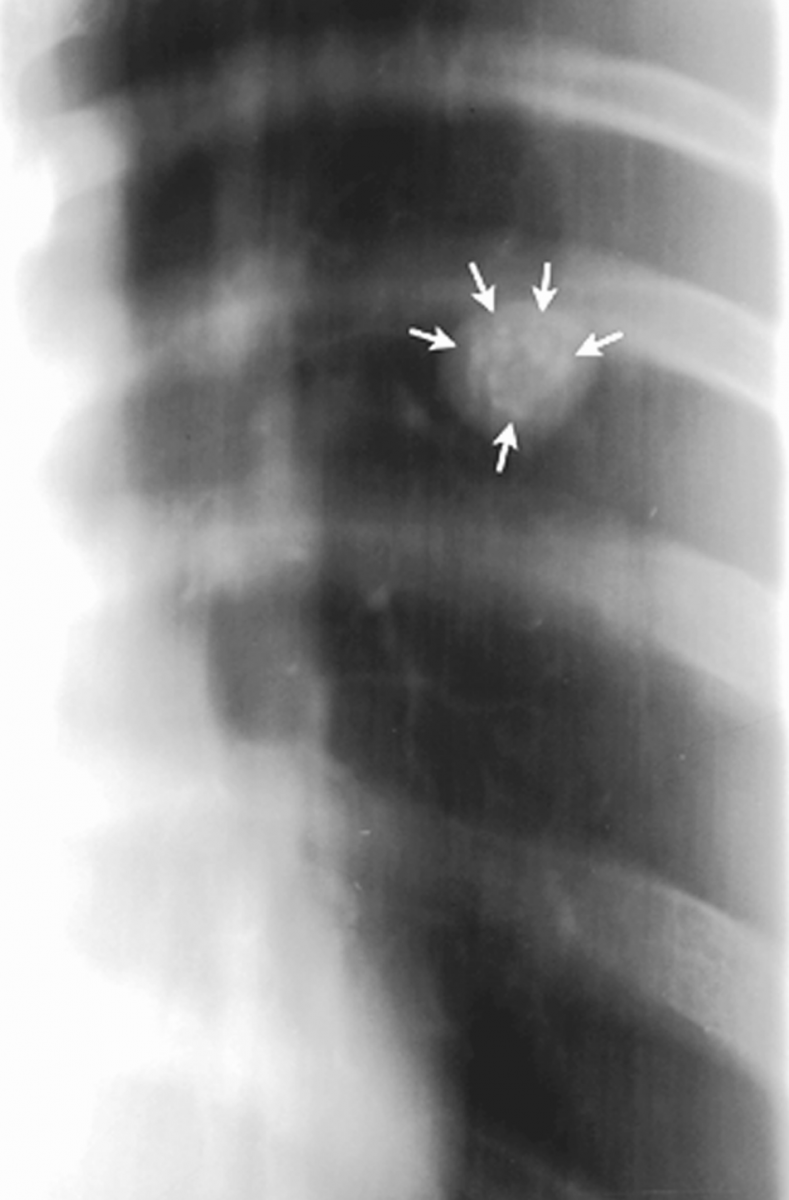
Figure 3 This enlarged linear tomogram displays the sharp margins of the lesion (arrows), as well as the dense calcifications in a popcorn configuration within it.
Diagnosis
Metastatic tumor, amyloidoma, and any benign or malignant lung tumor were considered in this patient’s differential diagnosis. However, Ewing’s sarcoma was unlikely because there was no bone involvement,1 and a malignant lesion typically has a lobulated contour and an irregular or speculated margin with distortion of adjacent vessels.2 The so-called popcorn configuration also differed from the target-shaped calcification of conditions like histoplasmosis and the peripheral calcification of other granulomatous diseases, metastatic calcifications, pneumoconioses, pleural calcifications, and parasitic calcifications.
Discussion
A hamartoma is an overdevelopment of cartilage, fibrous tissue, blood vessels, or another tissue element that normally belongs at the site where it is found. Pulmonary hamartomas are benign lesions comprised of an abnormal mixture of epithelial and mesenchymal components. The lesions are generally more common in men and typically develop in the sixth or seventh decade of life in smokers.3 The majority of tumors are peripheral lesions in the parenchyma of the lungs, while endobronchial lesions are less common.4 Peripheral nodules are typically asymptomatic, and the lesions are discovered as an incidental finding on a chest radiograph.
Radiographically, a hamartoma appears as a sharply defined, rounded lesion of intermediate soft tissue density. Punctate areas of calcium deposit are contained within the sharply demarcated nodule, which varies from 2.5-9.0 cm in size when detected. Although hamartomas are rarely malignant,3 up to half of all solitary pulmonary nodules detected on conventional radiographs prove to be multiple nodules on computed tomography (CT), suggesting metastatic or granulomatous disease.5 Thin-section CT can also demonstrate patterns of calcification associated with different lesions, as well as fat density — a reliable sign of hamartoma.2,5
Take-home Message
In this case, an unsuspected solitary pulmonary nodule with parenchymal disease required differentiation of benign lesions from malignant ones in the most cost-effective manner. The simple enlargement of the nodule on a linear tomogram clearly demarcated distinct popcorn calcifications that strongly suggested a hamartoma, the most common benign pulmonary lesion that is often asymptomatic. Therefore, hamartomas can be found incidentally via chest radiograph, a cost-effective screening tool that should always be part of the initial evaluation in patient care.
References
1. Gladish GW, Sabloff BM, Munden RF, et al. Primary thoracic sarcomas. Radiographics. 2002 May-Jun;22(3):621-37. http://www.ncbi.nlm.nih.gov/pubmed/12006691.
2. Erasmus JJ, Connolly JE, McAdams HP, et al. Solitary pulmonary nodules: Part I. Morphologic evaluation for differentiation of benign and malignant lesions. Radiographics. 2000 Jan-Feb;20(1):43-58. http://www.ncbi.nlm.nih.gov/pubmed/10682770.
3. Gjevre JA, Myers JL, Prakash UB. Pulmonary hamartomas. Mayo Clin Proc. 1996 Jan;71(1):14-20. http://www.ncbi.nlm.nih.gov/pubmed/8538225.
4. Choh JH, Steimle CM, Ahmad Z, et al. Endobronchial hamartoma: a benign, obstructive pulmonary hamartoma. Contemp Surg. 2001;57:297-9.
5. Leef JL 3rd, Klein JS. The solitary pulmonary nodule. Radiol Clin North Am. 2002 Jan;40(1):123-43, ix. http://www.ncbi.nlm.nih.gov/pubmed/11813815.
About the Author
James D. Collins, MD, is Professor and General Radiologist in the Department of Radiology at the UCLA David Geffen School Medicine. He formerly served as Director of the required medical student training for the department and President of the James T. Case Radiologic Foundation. Collins has an extensive background of publications, consultations, and editorial positions, including his current post as the Radiology Editor for the Journal of the National Medical Association. He specializes in bilateral 3-dimensional MRI and MRA imaging of the brachial plexus and has been performing those studies since 1985.
