
A prespecified secondary analysis revealed no difference in male and female patients’ responses to tirzepatide in obesity-related HFpEF.

A prespecified secondary analysis revealed no difference in male and female patients’ responses to tirzepatide in obesity-related HFpEF.
Peters emphasized how the new findings reinforce treatment strategies for chronic rhinosinusitis with nasal polyps.

Bhatt discussed the latest data from a post-hoc analysis of the BOREAS and NOTUS trials.

Read about groundbreaking FDA decisions, exciting new heart failure research, and more.

This set of interviews collects perspectives from 11 leading clinicians in the heart failure field, cataloguing the biggest news from 2025.

Counting down the most impactful news at major medical meetings, it’s the HCPLive Five! This episode focuses on 5 key dermatology updates from EADV.
During this EADV interview, Lebwohl speaks to some of the most recent developments in the psoriasis research space.
Hanania discussed the rationale and findings of the BOREAS and NOTUS studies.

In this set of interviews, 6 leaders in the field of dermatology highlight their experiences at the 2025 EADV Congress.
Le discusses methods by which clinicians can ease the financial burden of obtaining and adhering to medication for their patients.

The investigative therapy from Windtree Therapeutics also maintained a favorable safety and tolerability profile versus placebo.
In this interview at EADV, Bernstein highlights dupilumab’s impact on urticaria activity in individuals with chronic spontaneous urticaria (CSU).

Explore the evolving landscape of heart failure endpoints, focusing on the significance of oral diuretic intensification in outpatient care.

Recent analyses detail the ongoing risk of hyperkalemia in heart failure patients, highlighting the importance of optimal medication dosing.

An analysis of data from the HELIOS-B trial indicated an overall reduction in GI events among the vutrisiran cohort compared to those receiving placebo.

100% of included participants were successfully defibrillated and exhibited 6-month improvements or stability in the majority of patients.

New 96-week data reveals aficamten's significant benefits for patients with nonobstructive hypertrophic cardiomyopathy.

Acoramidis significantly lowers cardiovascular event risk in ATTR-CM, emphasizing the need for early diagnosis & treatment.

Guideline-directed medical therapy and SGLT2i use has been associated with reduced total health care costs and a lower risk of hospitalization and death.
If this connection is proven, the gut microbiome may present an entirely new pathway for treatment and prevention of heart failure.

Patients taking multiple drugs simultaneously receive the same protective effect from finerenone, regardless of their degree of polypharmacy.

New research links marijuana use to higher readmission and mortality rates in heart failure patients.

Recent studies reveal finerenone's superior benefits over traditional MRAs in heart failure, showing reduced mortality and hospitalization rates.
This discussion highlights 24-week findings on povorcitinib for those with moderate to severe hidradenitis suppurativa (HS).
This EADV 2025 interview featuring Linda Stein Gold, MD, highlights her team’s findings on icotrokinra versus placebo and versus deucravacitinib in psoriasis.
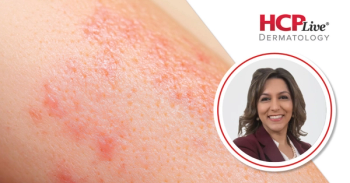
This interview at EADV 2025 features Mona Shahriari, MD, who highlighted recent findings on the drugs icotrokinra and povorcitinib in dermatology.
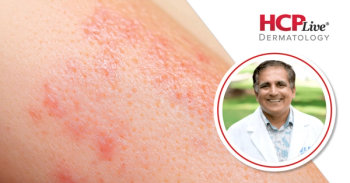
This interview highlighted findings on actinic keratosis following treatment with tirbanibulin.

A compiled list of the 8 most exciting presentations scheduled for the Heart Failure Society of American Annual Scientific Meeting 2025.

A compilation of 14 trials presented as late-breaking data at EADV 2025 Congress.

New findings from the POSITIVE trial highlight tildrakizumab's effectiveness in enhancing quality of life for psoriatic disease patients.