
Kadosh and Gorodeski discuss 2 of the major challenges facing both patients and clinicians dealing with HF and how to address them.

Kadosh and Gorodeski discuss 2 of the major challenges facing both patients and clinicians dealing with HF and how to address them.

New data suggest those who exhibit minimal early response to ruxolitinib cream may still achieve repigmentation with prolonged use extending to 2 years.

At AAAAI 2026, Nowak-Wegrzyn presented FPIES diagnosis in infants, emphasizing clinical recognition and updated oral food challenge protocols.

Stay updated with the latest healthcare breakthroughs, FDA actions, and new trial data, in this week’s essential news roundup.

The FDA approval of milsaperidone and a pair of positive trials in depression signal a month of progress in psychiatry.

Maddox discusses the AHA’s recent statement indicating that, should current trends continue, 6 in 10 US women will have cardiovascular disease by 2050.

Experts outline a 2026 vision for glomerular disease care, including new endpoints, combination therapies, and gene therapy advances.

The PARASOL initiative is reshaping glomerular disease trials through shared data and improved surrogate endpoints.

PLA2R antibodies are advancing precision care in membranous nephropathy and informing new surrogate endpoints through PARASOL.

Anti-nephrin autoantibodies are redefining minimal change disease and enabling more precise, steroid-sparing treatment strategies.

APOL1 kidney disease highlights the need for community trust, increased screening, and stronger clinical trial participation.

Rapid advances in glomerular disease, from single-cell tech to biomarkers and phase 3 trials, are reshaping diagnosis and treatment.

Phase 2 ALPHA-SOLAR data presented at AAAAI 2026 show navenibart provides >95% median attack reduction in HAE with favorable safety for 3- and 6-month prophylaxis.

This FDA News Month in Review provides a round-up of regulatory decisions from February 2026.
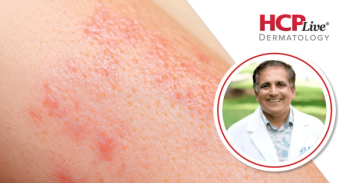
This Q&A with Neal Bhatia, MD, features a discussion conducted regarding GLP-1 use in patients with psoriasis and obesity or overweight.

In this segment of Liver Lineup, experts discuss the complexity of hepatitis B guidelines and why it’s important to empower clinicians and expand care beyond specialists.

This month in review features 7 allergy headlines in February 2026: FDA's decision on Dupilumab for AFRS, late-breaking data presented at AAAAI, & more.

Diabetes Dialogue cohosts Diana Isaacs, PharmD, and Natalie Bellini, DNP, discuss topline results from the TRIUMPH-4 trial, demonstrating retatrutide's efficacy in promoting weight loss.

Twice-daily delgocitinib cream 20 mg/g in those with moderate to severe chronic hand eczema led to significant HRQoL improvements.

Shamji discussed the advantages of PQ grass over conventional immunotherapy at AAAAI 2026.

A randomized crossover study shows that most patients prefer intranasal epinephrine for anaphylaxis due to its ease of use, portability, and the absence of needles.

Diabetes Dialogue cohosts Diana Isaacs, PharmD, and Natalie Bellini, DNP, cover the recently published trial results and what they mean for Eli Lilly’s newest GLP-1 molecule.
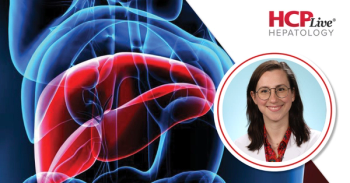
New research suggests MASLD risk may develop early in life, highlighting the impact of parental factors and cumulative childhood adiposity.

In the final part of this 6-part HCPLive Special Report, experts discuss the recently released HFSA/AAHFN joint statement on tech integration in heart failure care.

In part 5 of this 6-part HCPLive Special Report, experts discuss the recently released HFSA/AAHFN joint statement on tech integration in heart failure care.

In part 4 of this 6-part HCPLive Special Report, experts discuss the recently released HFSA/AAHFN joint statement on tech integration in heart failure care.

In part 3 of this 6-part HCPLive Special Report, experts discuss the recently released HFSA/AAHFN joint statement on tech integration in heart failure care.

In part 2 of this 6-part HCPLive Special Report, experts discuss the recently released HFSA/AAHFN joint statement on tech integration in heart failure care.

In part 1 of this 6-part HCPLive Special Report, experts discuss the recently released HFSA/AAHFN joint statement on tech integration in heart failure care.

COMBINE trial data show dupilumab added to omalizumab-assisted OIT improved desensitization and reduced GI symptoms, despite no sustained response benefit.