
The FDA has approved RNA interference therapeutic plozasiran for the treatment of familial chylomicronemia syndrome.

The FDA has approved RNA interference therapeutic plozasiran for the treatment of familial chylomicronemia syndrome.

Badimon explains the impact of sotagliflozin’s improvements in left ventricular mass, diastolic function, and 6-minute walk test results in patients without diabetes.

New qualitative findings reveal the underrecognized psychosocial burdens facing children with HAE and the unmet needs shaping their care.

Dual BLyS/APRIL inhibition with telitacicept led to significant 24h-UPCR reductions, lower progression-risk thresholds, and stable eGFR compared with placebo.

This November 18 episode explores early detection, diagnosis, and management of scarring alopecias and hair-care–related hair loss in patients of color.

Marston discusses the CORE and CORE2 trials, highlighting olezarsen’s success in lowering fasting triglyceride.
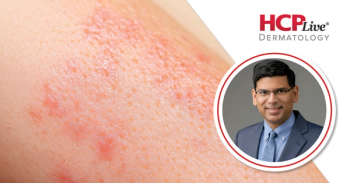
A phase 3b study in patients with skin of color found lebrikizumab provides reliable efficacy and safety for treating moderate-to-severe AD.

New ACAAI 2025 data show lebrikizumab improves disease control, quality of life, and work productivity in patients of skin of color with moderate to severe atopic dermatitis.
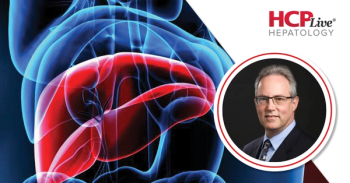
Data presented at AASLD 2025 show patients without severe neurologic involvement can achieve cognitive improvement with early detection and therapy.

These topline results from the ADHAND trial highlight tralokinumab’s use in adults with atopic dermatitis with moderate-to-severe hand involvement.
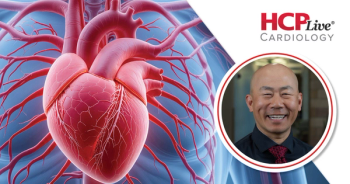
Le discusses the increased risk of mortality and major adverse cardiovascular events in patients with both PAD and AF.

The FDA acceptance of Arcutis's supplemental New Drug Application (sNDA) was announced for roflumilast cream 0.3% (Zoryve).

Data from 4 of the most impactful trials presented at the American Heart Association Scientific Sessions 2025.
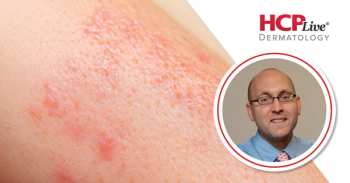
This Fall Clinical Dermatology Conference interview features Peter Lio, MD, discussing takeaways from several of his sessions at the meeting.

Lau discusses the use of virtual shared medical appointments for increasing access to cardiovascular care in a traditionally underserved population.

The FDA adds a boxed warning to delandistrogene moxeparvovec-rokl (ELEVIDYS) restricting indications to ambulatory patients with DMD.
Vogelmeier discusses major changes to the annual COPD report, including a broader threshold for escalating treatment after exacerbation.

Here, we spotlighted 4 late-breaker studies featured in our ACAAI 2025 coverage, along with 2 phase 3 trials.
After his sessions on TYK2 Inhibitors for psoriasis and on palmoplantar pustulosis, Vender discussed several key insights in this interview.
COPD is not a complex disease, Polverino explains. It's only complex to treat because of limited understanding.
In this Q&A, Soong discusses phase 2 results on povorcitinib, a JAK1 inhibitor that provides rapid relief from chronic spontaneous urticaria symptoms.
Benjamin Locksin speaks in this interview about his session titled ‘Encapsulating Progress With New and Emerging TYK2 Inhibitors for Psoriasis.’

Acoramidis also increased serum transthyretin in patients with wild-type ATTR-CM or the pathogenic variant ATTRv-CM.

FDA clears BrainsWay's Deep TMS for adolescents aged 15-21 years with major depressive disorder (MDD), expanding its use.

Polverino stresses the need for prospective analysis to validate CC16 and mucus plugs as viable biomarkers for COPD status.
Bakhos explains the ideal patient population and strategies inherent to LVRS success.
Friedberg explains how an emphasis on physical fitness prior to and after surgery can significantly better long-term outcomes.

Friedberg discusses the inconsistent clinical pathways from disease progression to intervention in patients with a pair of highly deadly lung diseases.

Cohosts Stephen Greene, MD, and Muthiah Vaduganathan, MD, sat down with Ambarish Pandey, MD, to discuss the revolutionary POLY-HF trial and how polypills can fit into heart failure treatment.
Sands explains the potential benefits of obefazimod and reviews findings from ABTECT 1 and ABTECT 2 presented at ACG 2025.