
Prothena halts birtamimab development after missing key Phase 3 trial endpoints for AL amyloidosis, prompting workforce reductions and cost-cutting measures.

Prothena halts birtamimab development after missing key Phase 3 trial endpoints for AL amyloidosis, prompting workforce reductions and cost-cutting measures.

With the priority review acceptance of the Biologics License Application, the FDA has assigned a PDUFA target action date of November 28, 2025.


In this interview, nerandomilast’s safety profile in idiopathic pulmonary fibrosis (IPF) and progressive pulmonary fibrosis (PPF) is highlighted.

Discover the critical role of Lp(a) measurement in assessing cardiovascular risk and its potential impact on CVD prevention strategies.

Hosts break down their biggest takeaways and detail their favorite sessions from the AACE 2025 annual meeting.

Recent results have confirmed faricimab’s efficacy, although a small sample size and short follow-up period invite further studies.

New predictive models enhance clinical decision-making for children with pneumonia, identifying severity levels to improve treatment outcomes effectively.

Real-world evidence highlights baricitinib's effectiveness and safety for treating atopic dermatitis and alopecia areata, showing high patient satisfaction and significant improvements.

Attaway reviews data demonstrating the potential benefit of a subspecialty clinic for overcoming current diagnostic and treatment-related challenges for sarcopenia in COPD.
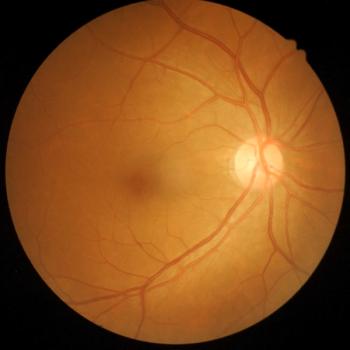
Type 2 macular neovascularization, reductions in outer nuclear layer and central foveal thickness, and intraretinal fluid may indicate higher odds of retinal atrophy.

This interview highlights phase 3 trial data on nerandomilast’s use in idiopathic pulmonary fibrosis (IPF) and progressive pulmonary fibrosis (PPF).

Despite 2020 guidelines discouraging first-generation antihistamines, ED diphenhydramine use for anaphylaxis and urticaria remained unchanged since 2019.

Prevot describes her research exploring the relationship between OSA severity and abdominal obesity metrics as well as changes in both metrics following CPAP.

Elizabeth Hale, MD, discusses the need to improve awareness and diagnosis of actinic keratosis.

Stay updated with the latest healthcare breakthroughs, including FDA approvals and innovative treatments, in this week's essential news roundup for professionals.

Panretinal photocoagulation and anti-VEGF therapy combined also results in better BCVA and were less likely to require additional treatment within 1 year.

These data from ATS 2025 highlight patients’ real-world outcomes related to treatment of their chronic obstructive pulmonary disease (COPD) with dupilumab.

The FDA approves treprostinil inhalation powder, enhancing treatment options for patients with pulmonary arterial hypertension and interstitial lung disease.

A recent study validated an online tool’s ability to accurately predict peanut allergy; the basophil activation test was the top performer, especially when paired with Ara h 2-sIgE.
While aflibercept 8 mg and faricimab both trigger intraocular pressure, the effects are transient and no more significant than other anti-VEGF treatments.

The analysis of pooled data from BOREAS and NOTUS highlight dupilumab’s sustained impact on lung function in patients with COPD and type 2 inflammation.

In this interview, Chalmers highlights the implications of his team’s subgroup analysis findings in the ASPEN trial, as well as unmet needs for non-cystic fibrosis bronchiectasis.

A new study identifies GDNF, IL-18R1, and IL-15RA as key biomarkers in seasonal allergic rhinitis, linked to symptom severity and immune pathways.

Triamcinolone acetonide injections effectively prevent diabetic macular edema after vitrectomy, highlighting its potential in postoperative care.

Hosts Kimberly Brown and Nancy Reau break down 4 of the top abstracts presented at the 2025 European Association for the Study of the Liver (EASL) Congress.

The FDA approves mepolizumab as a new maintenance treatment for COPD, showing significant reduction in exacerbations for specific patients.

Gyre Therapeutics plans to file an NDA in China based on the phase 3 CHB data and, pending IND clearance, is looking to initiate a phase 2 MASH fibrosis trial in the US.

A new study found ocular allergy affects 49.3% of students aged 11–18 years in Trinidad and Tobago—greater than in the US, Sweden, Brazil, and Uganda.

Discover innovative treatments for plaque psoriasis that cater to diverse skin tones, offering effective relief without the risk of discoloration.