
This approval of roflumilast foam as a once-daily topical treatment for psoriasis of the scalp and body followed the phase 3 ARRECTOR study findings.

This approval of roflumilast foam as a once-daily topical treatment for psoriasis of the scalp and body followed the phase 3 ARRECTOR study findings.

Calprotectin emerges as a promising biomarker for predicting atherosclerotic cardiovascular disease risk, independent of traditional risk factors.

Findings from a post hoc analysis of the phase 3 DIRECTION study highlight tezepelumab’s consistent benefit on asthma exacerbations across trigger categories.

Trief and MD candidate Justin Cheong physically peeled the Haab’s striae from a patient’s eye, allowing for better adherence and maintenance of the DSEK graft.

Susvimo is the first FDA-approved continuous delivery treatment for diabetic retinopathy capable of maintaining vision through one refill per 9 months.

Jain discussed Rejoyn, the FDA-approved digital therapeutic app that enhances major depressive disorder treatment with engaging brain training and mindfulness exercises.

At ATS 2025, these data highlight a potential connection between asthma development and prenatal exposure to microplastics and other forms of pollution.

Murphy discusses innovative surgical techniques for managing glaucoma and cataracts, emphasizing minimally invasive approaches and the future of ophthalmology.

This interview highlights the subgroup analysis findings of the ASPEN trial, specifically on brensocatib for non-cystic fibrosis bronchiectasis.

This special episode of Lungcast features Harold P. Wimmer, CEO of the ALA, and Francesca Polverino, MD, PhD, an ALA spokesperson.

Gerard Criner, MD, reviews findings from a secondary analysis of the phase 3 MATINEE trial of mepolizumab and looks ahead to the potential significance of the drug, if approved.

Only 50.3% of physicians consistently manage drug hypersensitivity. Study urges mandatory training, better protocols to improve patient safety.

In her interview at ATS, Willen further described elements highlighted in her presentation on new treatments for sickle cell disease.

This interview at ATS 2025 features a discussion of a ‘Pediatric Year in Review’ presentation on new therapies for sickle cell disease.

Stoller describes the potential of a novel AI model to help address AATD underrecognition, citing findings from his research on its performance in a Cleveland Clinic cohort.

Breazzano presented a video of a surgery repositioning an Akreos AO60 lens, highlighting the complicated nature of the procedure and its planning.

Early data on Selagine, Inc.’s immunoglobulin eye drops have indicated significant reductions in DED symptoms without differences in adverse events or tolerability.
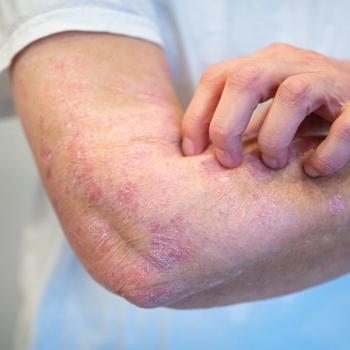
AC-201 at 25, 50, and 100 mg doses show promising phase 2 results, significantly reducing psoriasis severity.

The approval adds to Ritestar’s currently approved albuterol sulfate 2.5 mg/3 mL (0.083%), 1.25 mg/3 mL, and 0.63 mg/3 mL strength products.

High cow’s milk- and casein-specific IgE levels were key risk factors for reactions, including anaphylaxis, during low-dose oral food challenges in children.

Abramson discusses the potential benefits and limitations of treatment methods for Coats’ disease and further discusses the genetic component of its pathology.

This interview highlights the long-term, late-breaking results of the A DUE trial’s 24-month open-label extension on macitentan and tadalafil for PAH.

Criner reviews findings from AIRFLOW-3 and describes the subgroup of targeted lung denervation treatment responders being targeted in AIRFLOW-4.

In this episode, hosts explore real-world data on Beta Bionics' iLet automated insulin delivery system for diabetes management.

Results highlight dupilumab’s real-world benefit for asthma control in patients with and without coexisting chronic rhinosinusitis and/or nasal polyposis.

At ATS 2025, these post-hoc analysis findings on taladegib (ENV-101) highlight the treatment’s efficacy for idiopathic pulmonary fibrosis.

Attaway describes the importance of screening for and treating sarcopenia in patients with COPD and clinical gaps that complicate this process.

Veligrotug shows promising long-term Phase 3 results for treating thyroid eye disease, supporting its BLA submission in the second half of 2025.

In this interview at ATS 2025, Arthur Reingold, MD, highlights the role of pulmonologists and other health care providers in combatting vaccine misinformation.

The allergy medications of cetirizine or levocetirizine have led to pruritus in some patients who stopped using the medications after using them daily for a few months or years.