
Weeks discusses how tirzepatide is impacting treatment strategies for obstructive sleep apnea and driving collaboration across specialties.

Weeks discusses how tirzepatide is impacting treatment strategies for obstructive sleep apnea and driving collaboration across specialties.
Rubin reflects on the FDA approval of a SC induction regimen for guselkumab (Tremfya) in UC and explains the significance of having both IV and SC induction options in IBD.

A meta-analysis confirmed CARATkids as a reliable tool for assessing asthma and allergic rhinitis control in children aged 6–12 years.

A recent publication from the HFSA highlights obesity, chronic kidney disease, diabetes mellitus, and hypertension as major driving forces.
This EADV 2025 interview featuring Linda Stein Gold, MD, highlights her team’s findings on icotrokinra versus placebo and versus deucravacitinib in psoriasis.

The approval of the selectively targeted somatostatin receptor type 2 agonist is supported by data from the phase 3 PATHFNDR-1 and PATHFNDR-2 trials.

The extended indication includes children 1 to < 5 years of age with homozygous familial hypercholesterolemia.

FDA approval of tirzepatide for OSA sparks a surge in referrals as experts highlight weight loss benefits, symptom relief, and patient selection.

A trio of experts and a patient advocate discuss unmet needs and recent innovations in kidney transplantation, from xenotransplantation to continuous hypothermic machine perfusion.

Biolinq Shine aims to provide continuous glucose monitoring, as well as sleep and activity data, without requiring injections.

Despite the global prevalence of hypercholesterolemia, only 11% of children receive lipid screening, leaving many cases undiagnosed.
This interview at EADV 2025 features Mona Shahriari, MD, who highlighted recent findings on the drugs icotrokinra and povorcitinib in dermatology.
Neuen describes varying treatment effects with 4 different classes of IgAN therapeutics and future opportunities for personalized treatment.

This episode highlights the results of the ATTAIN-1 and STEP UP clinical trials, examining the potential of their respective GLP-1 RA subjects.
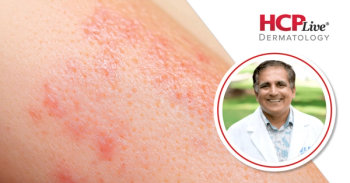
This interview highlighted findings on actinic keratosis following treatment with tirbanibulin.

CBT-I improved sleep outcomes and achieved 54% remission in patients with chronic pain, cancer, cardiovascular disease, and other chronic conditions.

Both ACOG and AAP have released statements disagreeing with the FDA label change for acetaminophen to reflect autism risk in children if used during pregnancy.

Investigators have highlighted diabetes and obesity as leading drivers of steadily increasing global cardiovascular mortality rates.
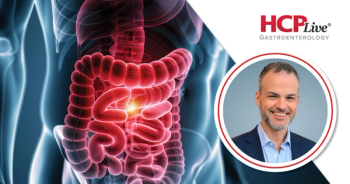
Staller describes unmet needs in gastroparesis and key recommendations for its diagnosis and management from the new AGA guidelines.

A compiled list of the 8 most exciting presentations scheduled for the Heart Failure Society of American Annual Scientific Meeting 2025.

Less than optimal outcomes with ASCVD also increased healthcare resource utilization and costs.

Despite prior trials indicating the possibility, the AUSPICE study failed to display any difference in risk between patients receiving the vaccine versus placebo.

In a study, the prevalence of transthyretin cardiac amyloidosis reached 17% in Black men over 75 years old.

Findings from an analysis of GBD 2021 data highlight increases in the absolute number of ASCVD cases and deaths among the working-age population.

AKINESIS study finds kidney tubule biomarker IGFBP‑7 predicts 1-year death and heart failure readmission in acute heart failure patients.

New research supports fimasartan, atorvastatin, and ezetimibe combination to blood pressure and cholesterol levels.

This Sickle Cell Awareness Month, HCPLive draws attention to recent advancements made in SCD treatment and how much still needs to be done to address it.

Findings suggest post-stroke dysphagia is associated with adverse psychosocial outcomes at 3 months, emphasizing the need for comprehensive rehabilitation strategies.

Parent company CeleCor Therapeutics announced positive safety and efficacy endpoint data for their investigative GPIIb/IIIa inhibitor versus placebo.

Although PREVENT was more consistent among the overall population, PCE saw more accurate readings in those taking statins within the trial period.