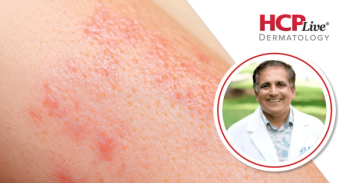
This segment of Bhatia's interview highlights imsidolimab's reported durability of response for those with generalized pustular psoriasis (GPP).

This segment of Bhatia's interview highlights imsidolimab's reported durability of response for those with generalized pustular psoriasis (GPP).
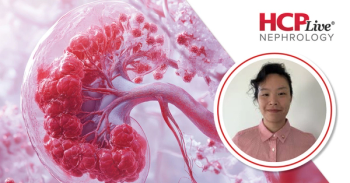
Yu, MD, Adler, MD, share their perspectives from a cohort study on in-sequence, and out of sequence paired kidneys transplants.

Long discusses AFFIRM data showing strong clinical and endoscopic outcomes with SC risankizumab (Skyrizi) induction in Crohn’s disease.

Wood discussed factors that go into shared decision making for multi-food allergy.
At AAAAI 2026, DiGiacomo discussed how artificial intelligence, genetic testing, and biomarkers could improve diagnosis and precision care.

Alexander discusses his recent study comparing 61 trials of 4 main GLP-1 RAs, highlighting their increased efficacy for weight loss in female patients.

In this episode, experts discuss solitary confinement, aging in prison, and gaps in care for incarcerated populations.

McWilliam discussed findings from the TreEat study at AAAAI 2026.

Salvatore Viscomi, MD on closing CKD health disparities through community testing, AI equity, and accessible care for underserved populations.

At AAAAI 2026, Torres highlighted phase 3 peach immunotherapy research, nanostructure preclinical models, and a phase 2 prebiotic trial for LTP allergy.

This interview highlights the significance of the potential FDA approval of imsidolimab for patients with generalized pustular psoriasis (GPP).

Kadosh and Gorodeski discuss the value of including non-specialists in HF care and the challenges of prescribing 4 pills in a time of increasing medical distrust.

At AAAAI 2026, Magerl discusses phase 2 data showing BW-20805 reduced hereditary angioedema attacks by up to 100% with dosing intervals of up to 6 months.

Satish discusses his recent study highlighting significant increases in first AMI in-hospital mortality among young adults, particularly in women.

These data, resulting from the INTEGUMENT-OLE trial, highlight the efficacy and safety of roflumilast cream (ZORYVE) 0.05% in children aged 2 - 5 years.

The approval comes after the FDA requested GSK submit an sNDA for leucovorin for the treatment of CFD-FOLR1.

This March episode highlights one SOCS member’s journey in dermatology, covering clinical and research passions as well as lessons learned along the way.

Wood discussed the latest long term data from OUtMATCHat AAAAI 2026.

Budini discusses his recent study investigating the trajectory of weight regain after GLP-1 cessation, given current high discontinuation rates.

February 2026 saw new data and expert interviews from AAAAI 2026.

The FDA accepted Mineralys Therapeutics’ NDA for lorundrostat in hypertension, but the selective aldosterone synthase inhibitor fell short in a phase 2 obstructive sleep apnea trial.

Phase 3 RAINIER exhibits positive results for povetacicept in adults with IgA Nephropathy in a 36 week interim analysis.

Having a primary care provider may be associated with reduced emergency department utilization for patients with ESKD receiving dialysis, according to new research.

A trio of experts discuss how simplifying hepatitis B guidance could help more clinicians diagnose, treat, and support patients living with chronic infection.

At AAAAI 2026, Larenas-Linnemann discusses evidence, safety, and long-term benefits of sublingual immunotherapy for allergic rhinitis and asthma.

Williams explains colorectal cancer screening options, persistent barriers to uptake, and strategies to improve screening rates.

An economic model suggest using lower quality, but acceptable kidneys for older adults may be cost efficective and clinically valuable.
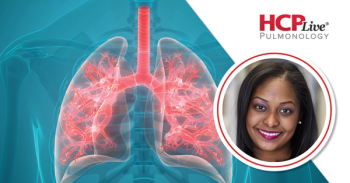
Using race-neutral spirometry equations such as GLI 2012 and GLI 2022 may improve diagnostic accuracy and reduce disparities in lung disease assessment.

Bimekizumab was assessed using the International Hidradenitis Suppurativa Severity Score System (IHS4) outcomes in the BE HEARD I and II studies.

Kadosh and Gorodeski discuss 2 of the major challenges facing both patients and clinicians dealing with HF and how to address them.