
ATTD 2026 reveals AI “digital twins,” next-gen closed-loop pumps, and sensors that boost time-in-range and ease diabetes management.

ATTD 2026 reveals AI “digital twins,” next-gen closed-loop pumps, and sensors that boost time-in-range and ease diabetes management.
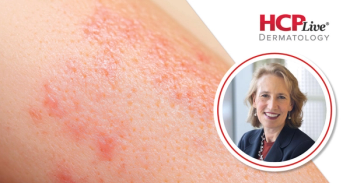
This Q&A highlights Dr. Alexa B. Kimball's responses to questions on the FDA approval of secukinumab for pediatric patients with hidradenitis suppurativa (HS).
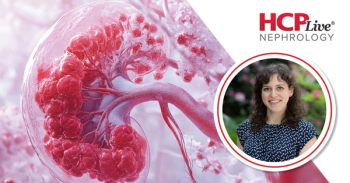
Data from 39 kidney biopsies may challenge longterm beliefs about diabetic nephropathy in CKD.

In a rapidly evolving therapeutic landscape, how can clinicians make decisions to personalize IgAN care?

The MiniMed Flex is a discreet, smartphone-controlled insulin pump that is roughly the size of 2 stacked insulin vials.
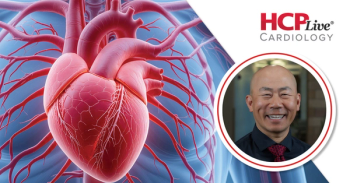
Le discusses the AHA/ACC clinical guidelines for the management of dyslipidemia, highlighting the shifting attitude towards hypertriglyceridemia treatment.

This discussion reflects on great speakers, the unique pressures women face on the podium, and what it takes to build a national platform in academic medicine.

BrainsWay’s SWIFT protocol reduced treatment visits from ~36 to 6 while maintaining response and remission rates in a randomized multisite trial.

FDA clears once-daily icotrokinra for moderate-to-severe plaque psoriasis in adults and teens.

Should clinicians consider neighborhood disadvantage the same way they measure proteinuria and GFR for kidney health?
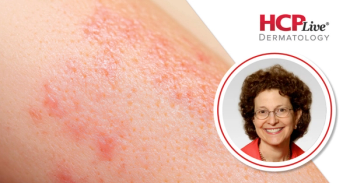
Following the release of new results from the phase 3 ADorable-1 trial on lebrikizumab in pediatric patients, Amy Paller, MD, speaks in a new Q&A interview.

Le discusses the AHA/ACC clinical guidelines for the management of dyslipidemia, highlighting the return of LDL-C target goals for ASCVD prevention.

High costs, limited insurance coverage, and adherence challenges remain key barriers to broader SLIT use in the US, Larenas-Linnemann says in an interview at AAAAI 2026.

KDIGO Launches Focused Update to 2024 CKD Guideline on Emerging Therapies

Allocating lower but acceptable quality kidneys to high-risk patients could introduce equity.

This segment of the latest episode of The Medical Sisterhood highlights insights into the career of Natasha Mesinkovska, MD, PhD.
At AAAAI 2026, Ramsey discussed the shift toward race-neutral spirometry equations, their clinical implications, and barriers to widespread adoption.

The FDA has issued its third CRL to topical ocular reproxalap for the treatment of signs and symptoms of dry eye disease.

Even Routine Urinalysis Can Reveal Hidden IgA Nephropathy, Especially in Young Adults

Le discusses the AHA/ACC clinical guidelines for the management of dyslipidemia, highlighting PREVENT and a renewed focus on Lp(a).
McDonald discusses new trial data suggesting an initial vancomycin taper may reduce early recurrence of C difficile infection.
This segment of Eichenfield's interview highlights long-term safety findings on roflumilast cream .05% for children 2 - 5 years old with atopic dermatitis.

New data show tegoprubart enabled insulin independence and strong glycemic control in adults with long-standing type 1 diabetes undergoing islet transplantation.

Allergy experts weigh ruxolitinib, tapinarof, and roflumilast—new nonsteroidal creams that reshape atopic dermatitis care beyond steroids.

This conference preview with Kathryn Berlacher, MD, MS, and Kristen Campbell, PharmD, highlights the top 6 trials to watch at ACC 2026.

Experts unpack biologics, molecular know-how, and patient trust—plus key previews for the upcoming AAD Annual Meeting.

Investigational RNA interference therapy targeting prekallikrein enters accelerated regulatory pathway as phase 2 trial evaluates long-acting prophylaxis for HAE.

The FDA has set a PDUFA date of December 30, 2026.

Data from the ACCESS II trial, ACCESS extension study, and a body composition study provide an early look at efficacy and safety data of a new investigational oral GLP-1.

This interview highlights key points regarding new data on roflumilast cream 0.05% in children aged 2 to 5 years with atopic dermatitis.