
Interim phase 3 APeX-P data presented at AAAAI 2026 show sustained 48-week reductions in moderate and severe HAE attacks with oral berotralstat in children aged 2–11 years.

Interim phase 3 APeX-P data presented at AAAAI 2026 show sustained 48-week reductions in moderate and severe HAE attacks with oral berotralstat in children aged 2–11 years.

Catch up with FDA decisions, key trial results, and more.
Neal Bhatia, MD, speaks on new data on ixekizumab and tirzepatide for weight loss and psoriasis, as well as the implications of these data regarding GLP-1 use.
Nguyen discussed how her study findings can help alleviate inhaled corticosteroid hesitancy for families.

Early gut microbiome changes in c-section infants linked to food sensitization at 12 months, highlighting a potential critical intervention window.

Trial data show early microbiome shifts after vaginal microbial transfer in cesarean-delivered infants, with differences in food sensitization at 12 months.

Nguyen emphasized the safety of inhaled corticosteroids vs SABA-only therapy in pediatric asthma.
HLA-A*32:01 carrier screening may identify patients at elevated risk for lamotrigine-induced eosinophilia and systemic symptoms, Krantz says at AAAAI 2026.

Prospective case-control data presented at AAAI 2026 demonstrate a 12-fold increased odds of DRESS among lamotrigine-treated HLA-A*32:01 carriers.

On Rare Disease Day 2026, clinicians share their perspective on the evolving landscape for IgAN and C3G.

Families and clinicians highlight the race against time in biliary atresia, where early recognition can change the course of a child’s life.
Retrospective analysis presented at AAAAI 2026 found 12%–15% risk reductions in asthma exacerbations over 3 years among patients without diabetes with overweight and obesity.
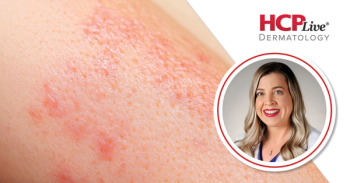
Once-daily VTAMZA delivers week-one sleep gains and lasting itch control in atopic dermatitis, with safety consistent across studies.

Post hoc pooled data from ADORING 1 and 2 demonstrate statistically significant improvements in patient and family sleep scores in children aged 2–17 years

Sicherer discussed how findings from CAFETERIA may inform treatment decisions for children with high-threshold allergy.

Anadani discusses a recent study highlighting the myriad contributing factors in the steadily increasing risk of maternal stroke across the country.

Brown discusses a destigmatised future of obesity management in patients with kidney failure.

Bhatt discusses a recent study from GSK highlighting AREXVY’s efficacy in lowering major adverse cardiovascular events.

Pressman discusses his recent study highlighting the interplay between right ventricular end-diastolic volume and ejection fraction and the left ventricle’s health.
4-year LUCENT data show mirikizumab reduces UC-related hospitalizations and surgeries, supporting durable remission and potential disease modification.

In this interview, Neal Bhatia, MD, highlights several key points about treating psoriasis and obesity via ixekizumab and tirzepatide.

Expert panel identifies high agreement for multi-organ involvement, but uncertainty remains in moderate isolated skin or mucosal symptoms.
New data show a >10% drop in newborn hepatitis B vaccination, raising concern for rising chronic infection and future liver cancer risk.

This February episode of The Medical Sisterhood highlights key points on juggling work as a clinician and work as a mother.
Willingness to pursue living kidney donation is shaped by relationships, health, and a multidisciplinary approach, not just finances.

Creager discusses the accomplishments of the ACC and AHA’s joint PE clinical practice guideline while continuing to look to the future of the disease.
Hurtado-Lorenzo discusses trial data showing time-restricted feeding in Crohn’s disease results in weight loss, symptom improvement, and microbiome changes.

Jolley discusses the team’s combination of volume rendering and cardiac MRI-based valve evaluation to provide 4D models of the heart and blood flow.

Systematic review identifies patient and clinician barriers to obesity management before kidney transplantation, with Adrian Brown, PhD.

Updated HAE guidelines support early self-treatment; Cancian discuss transitioning patients from injectable on-demand therapy to oral sebetralstat after KONFIDENT data.