
Alcohol, stress, drug use, and not seeking help are all risk factors for chronic depression.

Alcohol, stress, drug use, and not seeking help are all risk factors for chronic depression.

CBD has shown promise in treating anxiety and psychosis.

There remains a need for better treatments for bipolar disorder when it is identified early.

Mixed feature depression can be difficult to diagnose because the symptoms can be similar to other psychiatric disorders.

There remains a need for more research into the connection between grief and depression following a death.

Borderline personality disorder remains one of the most difficult to understand conditions in psychiatry.

Esketamine is seen as a major breakthrough in finding more treatments for depression.

Dr. Shear talked about the similarities and differences between depression and grief and the importance of recognizing both.

Dr. Kushner discusses obesity as a risk factor for CVD and how preventive cardiologists can better target weight management to support patient health.

Over 60% of patients in the second year of TENAYA and LUCERNE were on 16 week dosing in the faricimab group.

Machine learning algorithms successfully predicted visual and anatomic outcomes and dosing frequency in patients with macular edema secondary to CRVO.

Data show 77% of eyes with DME in the IRIS Registry received only one type of anti-VEGF agent over an average follow-up of 2.3 years.

The port delivery system provides good visual outcomes for patients with nAMD despite presence of fluid in post-hoc analysis of Archway trial.

A post-hoc analysis of the GATHER1 trial suggests avacincaptad pegol reduced GA lesion growth across all distances from the foveal center point, when compared to sham.

Longer time to intravitreal aflibercept injection (>3 months) showed less visual improvement versus shorter time (<1 month).

Data show metformin was associated with reduced odds of developing AMD, with low to moderate doses having the greatest potential benefit.

Dr. Singh discusses the promise of durability with the agent and the potential for reducing the amount of patient visits, while preserving visual outcomes.
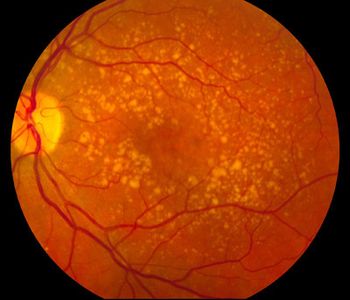
More than 60% of faricimab patients could be treated every 4 months at 2-year mark of TENAYA and LUCERNE studies.

Overall 70% of the patients in both PDS with ranibizumab and ranibizumab alone had almost no retinal thickness fluctuations.

The phase 3 data presented at the ISTH 2022 Annual Congress show an 86% reduction of treated bleeds in patients with hemophilia A or B with inhibitors.

Real-world patients showed improvement in visual acuity and anatomic parameters, but are still at an early time point in TRUCKEE study.

Evidence of pegcetacoplan treatment effect observed in the distribution of patients across quartiles in both OAKS and DERBY.

Monthly or every other month intravitreal pegcetacoplan reported to be tolerated in DERBY and OAKS trials.

The best overall outcomes were attained in eyes without fluid and ellipsoid zone integrity preservation, investigators said.

Travis Vandergriff, MD, discusses the unique collaboration with ophthalmologists and urologists in the rare skin condition.

An analysis of PSOARING 1 and 2 data show the topical therapy is efficacious across a wide spectrum of psoriatic disease and patient demographics.
An analysis suggests patients on the biologic fared better with adherence and outcomes than those on competitors including adalimumab.


At HiD 2022, Dr. Budoff discusses the use and utility of coronary calcium scoring for risk stratification

Dr. Martin explores the explosion of new consumer technologies and how to close the gap in disparities between data collection and using the data in practice.