
In this quiz, we examine your knowledge of biologic therapy for the treatment of CRSwNP.

In this quiz, we examine your knowledge of biologic therapy for the treatment of CRSwNP.

Positive 2-year results from the open-label compensated MASH cirrhosis arm of the MAESTRO-NAFLD-1 trial highlight resmetirom’s impact on liver stiffness.

On Feb. 26, 2025, Arcutis Biotherapeutics announced the FDA's acceptance of their sNDA for roflumilast cream 0.05% in AD for children aged 2 to 5 years.

Survey findings highlight pediatric nephrologists’ reluctance to list patients with kidney failure due to C3G or IC-MPGN for kidney transplantation.

Sebetralstat has a PDUFA date of June 12, 2025, for treating HAE in patients 12 years and older.

In this Diabetes Dialogue segment, hosts discussed the strengths of trial diversity and HbA1c reductions with automated insulin delivery in type 2 diabetes.

These data point to increasing omega-3 consumption as a potential preventive strategy against atopic dermatitis.

A novel fast-track protocol was linked to a lower incidence of delirium and reduced LOS after transplant without increasing rejection and readmission risk.

A new study presented at AAAAI 2025 linked pediatric obesity to a greater risk of developing allergic rhinitis, with a 1.24x increased risk at 1 year.

In this quiz, we test your knowledge of evidence-based use of intranasal corticosteroids for patients with CRSwNP.

The regulatory agency has set a PDUFA target action date of August 27, 2025, for the investigational gene therapy.

In this analysis, investigators sought evaluate the effectiveness and safety of adding low-dose oral minoxidil to the standard anti-inflammatory treatment of lichen planopilaris.

Despite decreasing death rates, an analysis of GBD 2021 data highlights the increasing incidence of cirrhosis and other chronic liver diseases.

Afif El-Hasan, MD of Kaiser Permanente in California joins host Albert Rizzo, MD, to discuss the acute effects of wildfire smoke on air quality.

Three subgroup analyses of the phase 3 NAVIGATOR study are set to be presented at the AAAAI/WAO Joint Congress.

Phase 3 ANCHOR-1/2 data support depemokimab as a twice-yearly treatment for CRSwNP, effectively reducing blood eosinophil counts over 52 weeks.
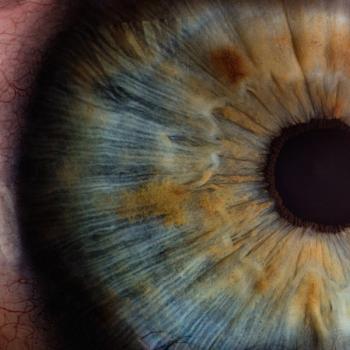
Eyelid pressure patching concomitantly with a decellularized, dehydrated amniotic membrane could prove a novel method to address PCED.

An audio recap of the top 5 stories in healthcare news from the week of 02/16 - 02/23.

These data suggest that psoriasis treatment algorithms may benefit younger patients if they implement a step-down rather than step-up approach to biologic use.

A real-world analysis found mepolizumab significantly improved symptoms in CRSwNP patients, reducing SNOT-22 scores, oral corticosteroid use, and surgery rates.

HCV screening in pregnant women increased following universal screening guidelines, but the proportion of women who were ever tested remained low.

Ahead of AAAAI 2025, HCPLive spoke with Jaroenpuntaruk about varying hypersensitivity reactions in patients with atopic diseases on biologics.

Epioxa has been assigned a target action date of October 20, 2025, for the regulatory agency’s review of the NDA.

These findings highlight the prevalence of atopic dermatitis among adults in France, as well as the different factors associated with various disease profiles.

FDA approves expanded use of buprenorphine extended-release injection for opioid use disorder, adding new injection sites and reducing treatment induction time.

Five years after peanut oral immunotherapy, children showed lower allergen-specific IgE and greater IgG4 levels than placebo, indicating sustained immune tolerance.

From 1990-2021, the ASIR, ASMR, and age-standardized DALY rate of chronic kidney disease increased.

The analysis of US healthcare claims data suggests noninvasive methods are being used more often than liver biopsies for the diagnosis of MASH.

These phase 3 findings from the INTEGUMENT-PED clinical trial highlight the efficacy and safety of roflumilast cream 0.05% for pediatric patients with atopic dermatitis.

Phase 3 WAYPOINT trial shows tezepelumab significantly reduces nasal polyp severity, congestion, and surgery needed in severe chronic rhinosinusitis with nasal polyp.