
In part 1 of 5, panel moderator gives an overview of the Special Report focusing on the approval of Xanomeline and tropism chloride capsules for schizophrenia.

In part 1 of 5, panel moderator gives an overview of the Special Report focusing on the approval of Xanomeline and tropism chloride capsules for schizophrenia.

Time-restricted eating, even without calorie restriction, improved glycemic control and cardiometabolic health in patients with metabolic syndrome.

Education and employment, social and emotional functioning, and healthcare access are notable factors influencing pain in sickle cell disease.

In the allergy news space, this Month in Review highlights some of the most significant stories covered by the editorial team.

In addition to the Breakthrough Therapy Designation, Boehringer Ingelheim announced the initiation of 2 phase 3 trials in adults with MASH and fibrosis and compensated MASH cirrhosis.

Findings highlight HCV infection burden among people without injection drug use, a known risk factor for HCV, and call for universal testing to better capture these individuals.

The biosimilars month in review highlights a new approval and 2023 cost savings

This retrospective analysis examined the current management of early and late anemia in a representative sample of the kidney transplant population in Spain.

These new data provide additional evidence for a range of benzene concentrations detected by in over-the-counter drug products with BPO maintained at room temperature.

A recent study shows the FDA's black-box warnings on antidepressants have led to decreased usage among youth, with an increase in suicide attempts and deaths observed.

This September 2024 month in review spotlights our top coverage of recent nephrology pipeline updates, guidelines, and research.

Anemia is frequently observed in primary care patients with HF and is linked to worse prognosis and comorbidities, including cancer.

The decision by the FDA follows an announcement by Biofrontera that there were no safety issues found following the submission of the medication’s supplemental New Drug Application.

In this Month in Review, we highlighted some of the most notable stories covered in the dermatology space during September 2024.

Test your knowledge of the phase 3 MAESTRO-NASH trial supporting resmetirom (Rezdiffra)’s FDA accelerated approval for MASH with this quiz.

Our September 2024 monthly recap in endocrinology spotlights regulatory updates, new guidelines, and the latest episodes of Diabetes Dialogue.

New data reports a notably heightened risk of atopic dermatitis in patients with prurigo nodulais compared with a control cohort.

In this latest edition of HCPLive’s monthly newsletter on recent US Food and Drug Administration (FDA) news, we highlight updates from across therapeutic pipelines.

A study of > 14,000 patients offers insights on real-world usage and characteristics of new rimegepant users, showing distinct patterns in acute vs. preventive treatment.

Patients taking GLP-1 RAs were more likely to show food retention and inadequate bowel preparation, even when the medications were held before the procedure.

A new post-hoc analysis has also revealed improvements in quality of life in patients’ caregivers.

Counting down the most impactful news at major medical meetings, it’s the HCPLive Five! This episode focuses on 5 key updates from Maui Derm NP+PA Fall 2024 in Nashville.
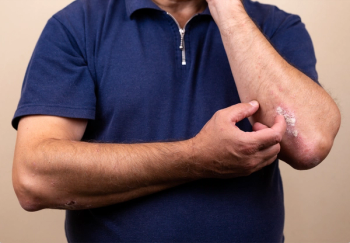
A study shows secukinumab clears skin lesions faster than ustekinumab in generalized pustular psoriasis, with improved responses observed by week 2.

A review of 5 highly anticipated FDA decisions expected by year-end, including acoramidis for ATTR-CM, olezarsen for FCS, sotagliflozin for T1D and CKD, and more.

Study findings highlight the individual and combined impact of smoking tobacco and drinking alcohol on achieving PASI75 after 8 weeks of treatment.

The approval is based on findings from BLUE-C, where Cologuard Plus demonstrated sensitivities of 95% for colorectal cancer and 43% for advanced precancerous lesions at 94% specificity.

An EADV 2024 study suggests dupilumab use during pregnancy is generally safe and may lower risks of preterm labor, gestational hypertension, and diabetes.

Crisugabalin at 40 mg/d or 80 mg/d significantly reduced daily pain in adults with postherpetic neuralgia, a phase 3 study found.

Further research is needed to understand the mechanism behind the link as antibiotic exposure is often necessary in care.
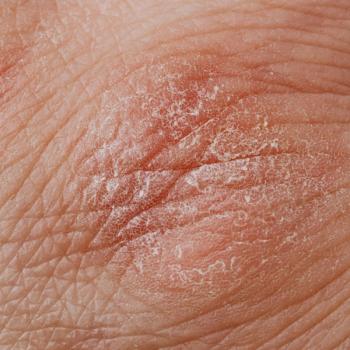
A small percentage of participants with psoriasis reported adequate adherence, with nearly half showing low adherence to phototherapy treatment.