
Schizophrenia experts discuss the stigma and misconceptions surrounding schizophrenia as well as current treatment options.

Schizophrenia experts discuss the stigma and misconceptions surrounding schizophrenia as well as current treatment options.

Presented at ASRS 2024, late-breaking RHONE-X data shows more than 90% of patients experienced the absence of DME after four years of faricimab.

Following this pooled analysis, there may be a need for additional research conducted to identify other targets for achievement of optimal repigmentation.

A study found children and young adults with cerebral palsy have more anxiety and less depression than those with a chronic condition or who are typically developed.

Primary care patients with MASLD who received a prescription for moderate- and high-intensity statins had a lower risk of progressing to a high-risk FIB-4 score.

Despite recommendations, an analysis of more than 3.2 million youths suggests fewer than 15% of children/adolescents receive lipid screening.

The FDA accepted Genentech’s supplemental BLA for Susvimo, based on the one-year results from the Phase III Pagoda and Pavilion studies.

This supplemental new drug application for once-daily roflumilast foam follows positive results of the ARRECTOR phase 3 study.

A study shows it is safer for nursing home residents on CYP2D6 opioids to take CYP2D6-neutral antidepressants than CYP2D6-inhibiting antidepressants.

Spigler provides insight into the development of new patient-focused guidelines for managing hyperkalemia in CKD and the importance of providing patients with such resources.
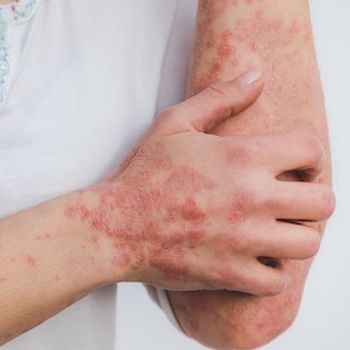
These findings represent the first comprehensive research into the greater frequency of APS autoantibodies linked with treating psoriasis with TNF inhibitors.

In this exclusive deep dive, a pair of leading dermatologists highlight the new data defining ruxotitinib cream, upadacitinib, and more agents from the revolutionary drug class.

An analysis using records from 40 million US patients highlights the surge in new GLP-1 receptor agonist use, with semaglutide dominating new prescriptions since 2020.

Eyes with GA that developed MNV in the GATHER trials experienced a slower growth rate with avaincaptad pegol than with sham treatment.

Data from INSPIRE and COMMAND were used to support risankizumab’s FDA approval for ulcerative colitis, demonstrating the IL-23 inhibitor’s impact on clinical remission.

Patients treated with PDS with ranibizumab refilled every nine months maintained vision improvements and experienced consistent safety over two years.

Johnson & Johnson submits a supplemental New Drug Application to the FDA for the approval of esketamine as a monotherapy for adults with treatment-resistant depression.

Bludorn joins Lungcast to review the increasing effects and signs of heat-related illness amid record-high temperatures.

The approval was based on the clinical, non-clinical, and analytical data proving eculizumab-aagh's bioequivalence to the reference drug.

These data suggest that the introduction of the concept of circadian syndrome contributes to greater understanding of risk management for those with psoriasis.

The phase 2 findings set up the foundation to design a phase 3 trial evaluating KP1077 for idiopathic hypersomnia.

The analysis of NHANES data found individuals with viral hepatitis and NAFLD as well as lower income and no college education were at a greater risk of mortality.

New research presented at ASRS 2024 links maternal asthma to a higher risk of advanced retinopathy of prematurity.

New data from the TEASE-3 trial presented at the ASRS meeting shows gildeuretinol acetate may blunt disease progression in early-stage Stargardt disease patients.

Over 2 years, the Port Delivery System demonstrated continued efficacy with fixed refill-exchange procedures every 24 weeks in the Phase 3 Pagoda trial.

Interim results from the PRISM population extension cohort found that 4D-150 was well tolerated and reduced the mean annualized anti-VEGF injection rate.

At 24 weeks, the oral therapy was well-tolerated and the safety profile remained in line with previous brepocitinib studies and the pharmacological class.
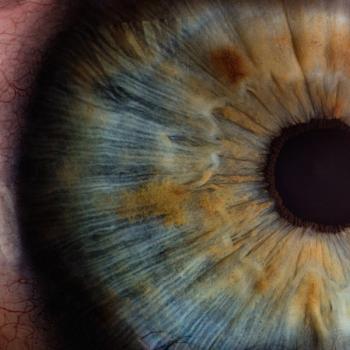
One-year 1/2 data examines the safety and efficacy of OCU400 gene therapy for the treatment of retinitis pigmentosa.

New data from RHONE-X at ASRS suggest over 90% of DME patients treated with faricimab (Vabysmo) were DME-free at 4 years, with nearly 80% on 3-4 month dosing intervals.

These data highlight results on efficacy, quality of life, and tolerability of DMF within routine clinical practice among those with plaque psoriasis.