
As part of our ARVO 2023 coverage, Dr. Sheth sat down with Pravin Dugel, MD, President of Iveric Bio to discuss avacincaptad pegol for geographic atrophy and a look back at Dr. Dugel's career and his transition into industry.

As part of our ARVO 2023 coverage, Dr. Sheth sat down with Pravin Dugel, MD, President of Iveric Bio to discuss avacincaptad pegol for geographic atrophy and a look back at Dr. Dugel's career and his transition into industry.

Siperstein highlighted some more major takeaways from her conference presentation, including facts about how to determine which patients are good candidates for microneedling.

In this interview, Siperstein explained some of the biggest takeaways from her conference presentation on advances in microneedling techniques, including radiofrequency specifically.

New phase 2b findings support the oral JAK1 inhibitor's capability to improve repigmentation versus placebo.

“These findings are exciting for us, and our overarching goal is to develop microbial biotherapeutics to treat food allergies," Mustafa Özçam says in an interview with HCPLive.

Symptoms including vestibular rashes and COVID toes have become less synonymous with SARS-CoV-2 infection. Why is that?

Speaking about one of his conference presentations, Chovatiya highlights the biggest takeaways regarding JAK inhibitors for alopecia areata, vitiligo, eczema, psoriasis, and more.

In another interview segment, Cohen described several other major takeaways from his conference presentation on psoriasis, comorbidities, and therapies.

In a conference interview, Cohen delved into the major takeaways covered in his presentation on the latest information related to psoriasis and its therapy.
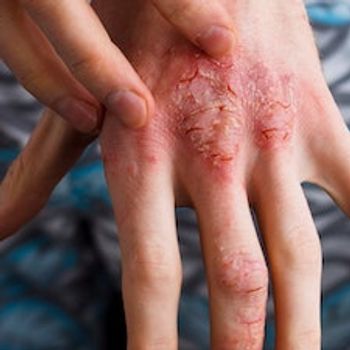
In recently-presented data, ivarmacitinib was found to have decreased severity of moderate-to-severe eczema compared to placebo.

As the pandemic passes its third year, one dermatologist foresees their field's own pursuit of specialty-specific vaccination guidelines.
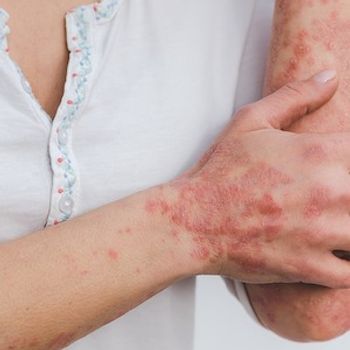
New late-breaking data showed that oral roflumilast treatment was both safe and efficacious against psoriasis, making it a potentially less expensive and convenient treatment alternative.

New late-breaking data supported prior results indicating that all primary and secondary endpoints were reached for the IL-4Rα antibody known as CBP-201 for atopic dermatitis patients.

New phase 2b data show the oral PDE4 inhibitor could reduce psoriasis area severity by more than 60% in 16 weeks.

New late-breaking data showed positive results for bimekizumab treatment of hidradenitis suppurativa, with higher response rates and a strong safety profile.

In research drawn from his conference presentation, Lim discussed the unique photoprotection needs of skin of color as well as some of the challenges faced by patients in this population.

A small molecule IL-17A inhibitor, izokibep has additionally shown efficacy in treating psoriatic arthritis—and will now be reviewed in a phase 3 HS trial.

Pediatric dermatologist Mercedes Gonzalez, MD, discusses the new phase 3 B-SIMPLE data supporting berdazimer gel as the first potential molluscum treatment.

Kwatra spoke about his team’s research into nemolizumab for prurigo nodularis in the OLYMPIA 2 trial, including the data and its implications for the future.

New data indicates that an oral TYK2 inhibitor known as NDI-034858 showed strong results for psoriasis patients in a recent phase 2b trial.

New data has indicated that patients with psoriasis who were not responding to ixekizumab or secukinumab responded well to risankizumab, an interleukin-23 inhibitor.

Late-breaking data on roflumilast cream for eczema patients in two phase 3 trials titled INTEGUMENT-1 and INTEGUMENT-2 showed strong results for the drug.

In a new interview, Eichenfield discussed the background of his presentation on late-breaking data from the INTEGUMENT-1 and INTEGUMENT-2 trials on roflumilast for atopic dermatitis.

New phase 3 data from the BRAVE-AA trial program showed some patients who had little response to initial baricitinib achieved hair growth in year 2 of treatment.

Late-breaking phase 3 data on nemolizumab monotherapy indicates substantial improvement in key primary and secondary endpoints for prurigo nodularis patients.

The Icahn investigator reviews new data suggesting ethnicity-based differences in molecular phenotype of atopic dermatitis, which may better inform treatment strategies for different populations.

Late-breaking data showed strong results for ruxolitinib cream as a treatment for vitiligo in the long-term extension of the TRuE-V1 and TRuE-V2 studies.

New phase 3 DELTA 1 trial data show the topical pan-JAK inhibitor doubled the rate of treatment success versus vehicle cream.

New late-breaking data showed positive results from the phase 2 trial on a novel septa-specific monoclonal antibody for HS patients.

Dupilumab showed strong results for uncontrolled moderate-to-severe hand and foot atopic dermatitis, meeting its primary and secondary endpoints in the trial.