
Birgit Vogel, MD discusses racial inequities and geographic variations in peripheral artery disease care in the United States.

Birgit Vogel, MD discusses racial inequities and geographic variations in peripheral artery disease care in the United States.

The investigators concluded that chronic psychological stress substantially impacts skin homeostasis and, consequently, skin aging.

Lawitz explains issues with traditional FXR agonists for MASH treatment and how FXR314 differs, potentially offering a more safe and efficacious option.

Milind Desai, MD describes favorable real-world safety outcomes from mavacamten use over 22 months of the Risk Evaluation and Mitigation Strategy (REMS) program.

HCPLive spoke with Kastl at NASPGHAN about the team’s study on the immune response to the COVID-19 vaccination among children with IBD.

Loomba reviews safety and efficacy data for the thyroid hormone receptor beta agonist from a phase 2a trial presented at AASLD The Liver Meeting 2024.

Ahmad Masri, MD, MS, discusses data from the FOREST-HCM trial examining effects of aficamten in patients with oHCM eligible for septal reduction therapy at baseline.
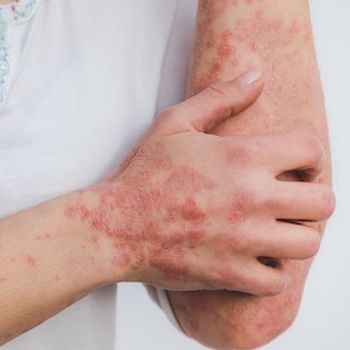
These retrospective, 52-week data highlight the efficacy and safety of bimekizumab for those with moderate-to-severe psoriasis.

Ahmad Masri, MD, MS, discusses a study from AHA 2024 detailing changing risk profiles and outcomes in trials among patients with ATTR-CM.

In this episode, hosts break down a pair of studies from AHA 2024, BPROAD and SUMMIT, as well as a study in JAMA projecting semaglutide eligibility in US adults.

Stephen Nicholls, MBBS, PhD, provides additional insight into KRAKEN trial data and the potential of muvalaplin.

Marianna Fontana, MD, PhD discusses Phase 1 interim results on the CRISPR-based gene-editing therapy, nex-z, for ATTR-CM.

This analysis evaluated the frequency of peanut-sensitized infants and toddlers being sensitized to other types of nuts.

Stephen J. Nicholls, MD, PhD discusses the robust efficacy and tolerability of zerlasiran for Lp(a) lowering, paving the way for Phase 3 development.

UCB has announced the FDA’s approval of bimekizumab, also known as Bimzelx, for patients with moderate-to-severe HS.

In these guidelines compiled by the ACAAI, the AAAAI, and the AAOA, recommendations are provided to insurance companies for documentation needed for allergen immunotherapy.

These data were announced by Protagonist Therapeutics, resulting from the 2 pivotal ICONIC-LEAD and ICONIC-TOTAL studies on the oral peptide icotrokinra.

At NASPGHAN, HCPLive spoke to Hamant about her team’s study that showed their algorithm was linked to increased VTE Prophylaxis among Pediatric IBD.

Barritt reviews real-world data about the use and benefits of GLP-1 RAs in patients with MASLD.

Bifidobacterium longum 35624TM reduces IBS symptom severity in children and adolescents, according to a study presented at NASPGHAN 2024.

Kowdley reviews open-label extension data for biochemical response, ALP normalization, and pruritus improvement from the phase 3 ELATIVE study.

At NASPGHAN 2024, HCPLive spoke with Duncan about how treating infants who appear to have reflux may actually result in greater repeat hospital visits.

Findings from the open-label extension of the ELATIVE study suggest sustained safety and efficacy for up to 3 years, with improvements in itch and sleep.

This segment of Han’s interview featured a discussion of the safety profile of dupilumab and ensifentrine, as well as other details in the GOLD 2025 Report.

Muthiah Vaduganathan, MD, MPH, discusses the need for cardiology to embrace a role for GLP-1 receptor agonists and incretin therapies, with a focus on obesity-related HFpEF.

Viet Le, PA-C, DMSc, breaks down new data and updates from AHA 24, spotlighting 3 specific trials and the applicability of artificial intelligence in care.

Muvalaplin, an oral treatment, reduced Lp(a) by up to 85% in a 12-week Phase 2 trial presented at AHA 2024.
Diabetes was associated with an increased risk of liver transplant or death and with liver-specific events in patients with PBC.

Marianna Fontana, MD, PhD describes the prognostic importance of a decline in eGFR in a retrospective cohort of patients with ATTR-CM.

Zhang explains the safety of different FDA-approved alcohol use disorder drugs in patients with alcohol-associated liver disease.