For patients with chronic liver disease, opioid prescriptions are common, and high-risk prescriptions are associated with hepatic decompensation, especially ascites.

For patients with chronic liver disease, opioid prescriptions are common, and high-risk prescriptions are associated with hepatic decompensation, especially ascites.
This rare leukemia form may be treated with ruxolitinib, which appears to be well-tolerated by patients.
Patients with myeloproliferative neoplasms (MPN) with essential thrombocythemia (ET) or polycythemia vera (PV) were found to benefit from hydroxyurea therapy.
Patients with myeloid/lymphoid neoplasms enrolled in the phase 2 clinical trial demonstrated pemigatinib was generally well-tolerated.
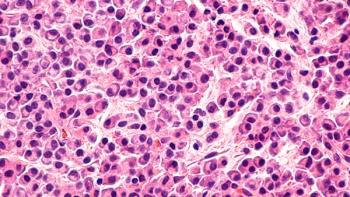
Nizar Bahlis, MD, highlights the phase 3 POLLUX trial evaluating daratumumab plus lenalidomide and dexamethasone for the treatment of patients with relapsed/refractory multiple myeloma.
ALD-related hospitalizations grew from 2008 to 2014, especially among women, Asians, Hispanics, and people 50 to 69 years of age.
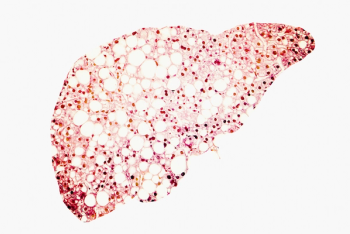
No studies to-date have looked at the combined risk of mortality for patients with thrombocytopenia and severe hepatic steatosis.
The leading etiologies for chronic-liver-disease-related-deaths over the last decade can be explained by NAFLD and alcoholic liver disease.

A new study confirms that HCV-RNA can be found in the nasal and rectal fluids of patients with high viral loads of HCV, raising the possibility of HCV transmission via these fluids.
A low dose of TDF may help to preserve renal function and maintain viral suppression in patients with chronic hepatitis B virus infection, even those with advanced liver disease.

Among the highlights, significant and sustained increases in hemoglobin without transfusions stood out.
Lead investigator, John Leonard, MD, reviews the phase 3 AUGMENT trial investigating lenalidomide plus rituximab for the treatment of relapsed/refractory follicular lymphoma.

Models show that for every gram of hemoglobin increased per deciliter, the risks associated with hemolytic anemia decrease.

Jorge Cortes, MD, highlights the phase 3 QuANTUM-R trial results and explains their clinical implications for patients with FLT3-ITD-mutated relapsed/refractory acute myeloid leukemia (AML).

After the therapy was approved by the FDA in May, investigators are anticipating its first real-world data will paint a clearer picture of its potential.
Patients’ daily bleed diaries helped investigators understand why some bleeds are treated, and some are not, as well as their causes.

Jasmine Zain, MD, recaps the current and upcoming treatments in T-cell lymphoma and highlights the key clinical trials playing a role in the rare disease.

The pediatrician discusses key differences between treating pediatric and adult patients with hemophagocytic lymphohistiocytosis (HLH) with the recently FDA-approved emapalumab.
The therapy attempts to restore fetal hemoglobin production.
Ravulizumab dosed every 8 weeks was noninferior to eculizumab given every 2 weeks, but the treatment did not achieve superiority.
Among the highlights observed in the data, the single-agent oral demonstrated significant improvement in progression-free survival (PFS).
Full results from the phase 3 MURANO trial showed a reduced risk in disease progression and 87.9% overall survival in relapsed/refractory chronic lymphocytic leukemia patients treated with venetoclax/rituximab.
Currently available anemia therapies only provide benefit for about half of patients with a lower-risk myelodysplastic syndromes (MDS)-related condition—and about one-quarter of such patients dependent on red blood cell transfusions.

Justin Taylor, MD, explains new and unique data that show cases in which a solid tumor can become a hematologic malignancy, or leukemia.
Males with myeloproliferative neoplasms (MPN) were found to have inferior survival compared to females in a well-defined, prospective, observational cohort.
The odds ratio of patients on the therapy reaching ≥33% RBC transfusion burden reduction was 5 times greater than those on placebo.
Long-term data shows familial haploidentical stem cell transplantation (HISCT) improves quality of life and neurocognition in sickle cell disease patients.

Cirrhotic patients with variceal bleeding were found to have greater morbidity in association with obesity.

How fatty liver disease and NASH—as well as the comorbidities associated with them—highlighted this year's discussions.

Asthma is the most common chronic disease of childhood. Cellphones might help improve its management.