Many trials for monoclonal antibody therapies for asthma reported small percentages of African American or Hispanic participants, and some didn’t report racial/ethnic demographics at all.

Many trials for monoclonal antibody therapies for asthma reported small percentages of African American or Hispanic participants, and some didn’t report racial/ethnic demographics at all.

Jay Edelberg, MD, PhD, of Sanofi and Robert Sanchez, PhD, of Regeneron shared about the recent ODYSSEY OUTCOMES trial of alirocumab (Praluent) and accompanying cost-effectiveness study.

The biologic AR101 was previously shown to significantly desensitize peanut allergies in a pediatric and adolescent patient population.
HRS-1 is a rare, acute complication of liver cirrhosis characterized by life-threatening and rapid progression that can end in renal failure. Terlipressin, a vasopressin analogue, could become the first therapy approved for the treatment of HRS-1 in the US.
Results may ease concerns for many physicians and patients who live in colder climates.

A 48-year-old man presents with severe atopic dermatitis that he’s had since childhood. Can allergy shots succeed where standard treatments failed?

The AR101 oral therapy—a biologic designed to desensitize peanut allergies, manufactured by Aimmune Therapeutics—is anticipated to be submitted for FDA approval in the next month.

Demystifying the oral food challenge starts with understanding its risks and benefits.

Study co-author Nicola Alexander Hanania, MD, explains what the new data means for practicing pulmonologists and allergists.

Busse explains the importance of having an additional treatment option and covers best practices for prescribing.

In 2017, roughly 5% of newborns had a peanut allergy. That’s a stark increase from the roughly 1% that characterized previous decades, according to researchers.

Researchers uncover the first report of cannabis allergy, suggesting that exposure to secondhand marijuana smoke may contribute to difficult-to-control asthma.

FF/VI met primary and secondary endpoints in increasing the mean proportion of days covered and time to index treatment discontinuation.

Researchers sought to balance the cost of autoinjectors with their lifesaving potential, and then boil the results down to an agreeable number. Here’s where they landed.

An updated epidemiological review paints a concerning picture of pediatric cow’s milk allergy in the US.

On the bright side: In-hospital education shown to be effective in helping bridge the gap.

Sterilizing or hand washing pacifiers doesn’t have the same allergy-suppressing benefits as sucking on them, according to new research.

In a real-world setting, omalizumab improved UAS scores, DLQI scores, response scores, and saw a similar or better safety profile.

Norah Terrault, MPH, MD, explains how the use of cannabis can affect a patient's status on the liver transplant list.

The therapy saw similar safety and efficacy profiles across the BORA, SIROCCO and CALIMA studies for the treatment of asthma.

Investigators have found that lowering LDL cholesterol may not be the only way to reduce cardiovascular events—reducing inflammation may be the key to improving outcomes.

Norah Terrault, MPH, MD, discusses gaps in treatment in people who are infected with hepatitis C virus and what steps clinicians should take when treating these patients.

The new approach could shave off time and costs from the current standard regimen.

Using relevant clinical trial data, investigators created a price tag of about $6300 annually for the very effective, often costly inhibitor.

James Januzzi, MD, discusses trial results from the PIONEER-HF trial and what cardiologists and hospitalists should know about sacubitril-valsartan.
At 12 weeks post treatment per protocol analysis with 8 weeks of glecaprevir/pibrentasvir treatment, patients with hepatitis C virus genotypes 1, 2, 4, 5 and 6 achieved a sustained virologic response.

Norah Terrault, MPH, MD, addresses the many unanswered questions and data surrounding cannabis use in liver disease.

Investigators praised the excellent effectiveness of both therapies, but advised care regarding the increased odds of adverse events requiring hospitalizations.

Steven Nissen, MD, discusses the updates to the cholesterol guidelines and how the risk calculator should not be considered an end-all be-all.
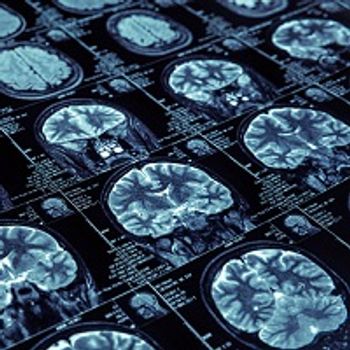
Investigators have found a pair of increased biomarker levels indicate patients with AF are at a greater risk of neurodegeneration.